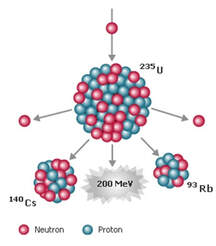
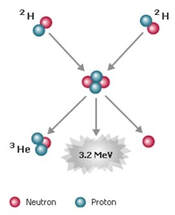
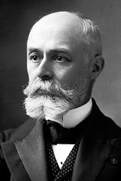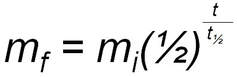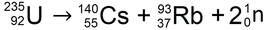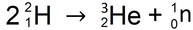KEY IDEA: Radioactive elements spontaneously release high-energy radiation through the conversions of mass and energy.
Nuclear Chemistry
|
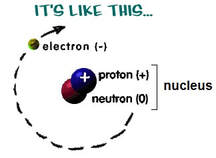
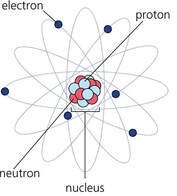
Nuclear reactions involve protons and neutrons within an atom’s nucleus.
(Chemical reactions involve electrons.) Some atoms have unstable nuclei and undergo nuclear changes in an attempt to get to lower energy, or "more stable." The atom may decay to form different atoms and in the process release incredible amounts of energy in the form of radiation, invisible and energetic particles emitted from the nucleus. Nuclear chemistry studies changes in the nucleus of an atom and explores the consequences of those changes. |
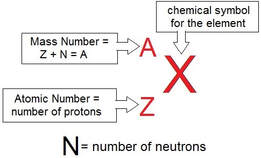
Recall isotopes:
Discovery of Radiation
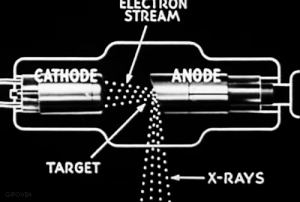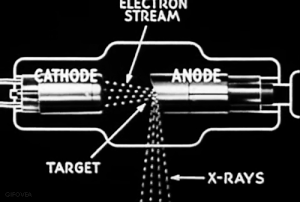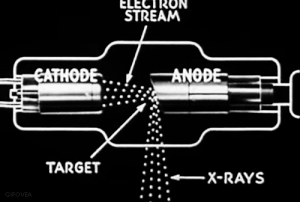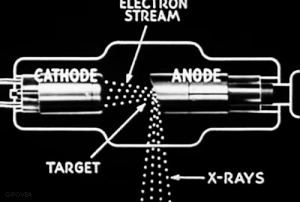
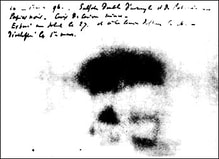
The discovery of radiation is a fortunate stroke of chance. Some put the birth of nuclear science at 1895.
Uncertain of what to call this this ‘mysterious’ radiation, Röntgen called them “X-rays," hoping to think up a more descriptive term once he learned more.
VIDEO Röntgen and the discovery of X-rays
|
1896 - 1st observation of radioactivity
Drawing on Röntgen’s work, French physicist Henri Becquerel attempted to prove X-rays were reemitted sunlight. When he stored a uranium salt on top of a photographic plate wrapped in black paper, surprisingly, he found the film had developed even though no visible light had reached it. His theory was false. The uranium salts emitted radiation on their own, not re-emitting stored energy from elsewhere. |
Becquerel demonstrated that the uranium rays resembled X-rays: highly energetic and ionizing. But unlike X-rays, "uranic rays" appeared spontaneously and were deflected by a magnetic field. And the intensity of the rays did not diminish over time.
|
1898 - discovery of radioactive elements
French-Polish physicist Marie Curie coined the term "radioactivity" to describe the spontaneous emission of radiation. Working with her husband Pierre, they figured a way to measure the intensity of the uranium rays and discovered other radioactive elements radium, polonium, and thorium. Marie concluded that the radiation originates from inside the uranium atom and an atomic phenomenon. The origin of the radiation was still a mystery because the existence of the nucleus was not known at the time. |
Becquerel had discovered radioactive decay products - small pieces of the uranium nucleus that had been ejected as it reordered itself into a different, more stable arrangement. For their joint discovery of radioactivity, Becquerel and the Curies share the 1903 Nobel Prize in Physics.
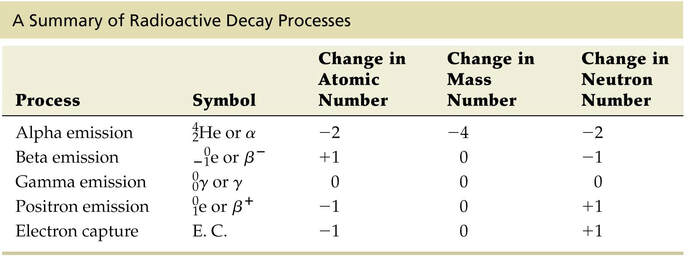
Research on radioactive elements from minerals led to the discovery of 3 distinct types of radioactive decay.
|
1902 - separation of radiation into alpha, beta and gamma.
Ernest Rutherford and Frederick Soddy explained radioactivity as spontaneous transmutations. Rutherford earned the Nobel Prize for his investigation into radioactive substances. He is credited as the father of nuclear physics and converted nitrogen into oxygen. |
Taken together, the work of Becquerel, the Curies, and Rutherford, represents the dawn of the nuclear age.
Investigate why radioactive decay occurs and the changes that occur in the nucleus in this simulation.
Nuclear StabilityThe nucleus of an atom contains positively charged protons and neutrons (with no charge) in a very tiny volume.
Stable nuclei can form despite the repulsion between protons thanks to an attractive force (strong nuclear force) supplied by neutrons. Unstable nuclei undergo spontaneous radioactive decay, releasing some form of radiation (gamma rays, electrons, or subatomic particles). |
All known combinations of protons and neutrons are summarized on the chart of nuclides:
Why some atomic nuclei decay or transform over time while others do not isn't entirely understood, but there are a few general trends that nuclear structure matters; stability is determined by a balance between protons and neutrons.
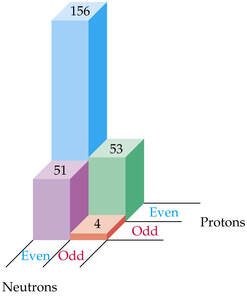
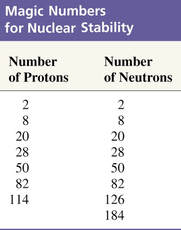
The existence of protons in pairs seems to have some stability on the nucleus. For protons, "magic numbers" are 2, 8, 20, 28, 50, or 82. These same numbers apply to neutrons.
Of all known stable isotopes, more than half have an even number of both protons and neutrons.
The existence of protons in pairs seems to have some stability on the nucleus. For protons, "magic numbers" are 2, 8, 20, 28, 50, or 82. These same numbers apply to neutrons.
Of all known stable isotopes, more than half have an even number of both protons and neutrons.
For lighter elements, the numbers of protons and neutrons is similar, but heavier elements have more neutrons than protons.
For "small" nuclei (less than 20 protons), a 1:1 ratio of protons to neutrons makes for a stable nucleus. As stable isotopes become larger, more and more neutrons are required to overcome the increasing repulsive force.
With more and more protons are packed in the nucleus, an excess of neutrons is required for stability.
For "small" nuclei (less than 20 protons), a 1:1 ratio of protons to neutrons makes for a stable nucleus. As stable isotopes become larger, more and more neutrons are required to overcome the increasing repulsive force.
With more and more protons are packed in the nucleus, an excess of neutrons is required for stability.
Useful links:
- Table of Nuclides for nuclear decay and mass data
- JANIS Nuclear Database to find any cross section for any nuclear reaction
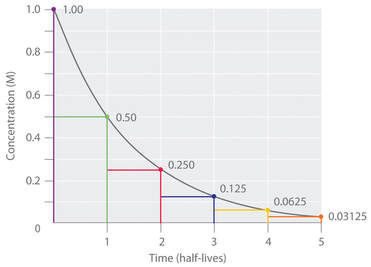
half-life
The decay of nuclei is governed by the laws of statistics. The period of instability varies from substance to substance at a known rate called a half-life – the time it takes for one-half the radioactivity to be emitted.
The half-life is constant, but is different for each isotope. Half-lives can vary from nanoseconds to billions of years.
The half-life is constant, but is different for each isotope. Half-lives can vary from nanoseconds to billions of years.
All nuclei with more than 82 protons are unstable. The presence of 83 protons in the nucleus tends to make it unstable regardless of the number of neutrons present. In fact, elements beyond uranium (92 protons) are so unstable that any amounts present when the Earth formed have long since decayed. Yet, unstable nuclei of any element can exist.
|
Half-lives are governed by the algebraic equation
|
where:
|
REVIEW
Explore Further
FUSION: Can we recreate the renewable power of stars down on Earth?
Modern Marvels: The Manhattan Project - Full Episode
Scientists, engineers, and the U.S. Army teamed up to detonate the first man-made atomic explosion. Dubbed the Manhattan Project, the top-secret undertaking was tackled with unprecedented speed and expense - almost $30-billion in today's money.
Scientists, engineers, and the U.S. Army teamed up to detonate the first man-made atomic explosion. Dubbed the Manhattan Project, the top-secret undertaking was tackled with unprecedented speed and expense - almost $30-billion in today's money.
Uranium: Twisting the Dragon's Tail
Born from the collapse of a star, uranium has brought hope, progress and destruction. It has revolutionized society, from medicine to warfare. It is an element that has profoundly shaped the past, will change the future and will exist long after humans have left the Earth.
Born from the collapse of a star, uranium has brought hope, progress and destruction. It has revolutionized society, from medicine to warfare. It is an element that has profoundly shaped the past, will change the future and will exist long after humans have left the Earth.