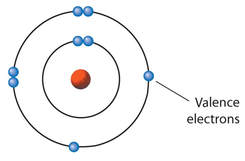
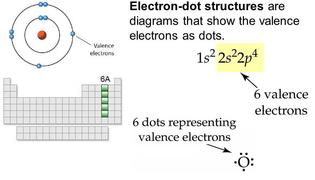
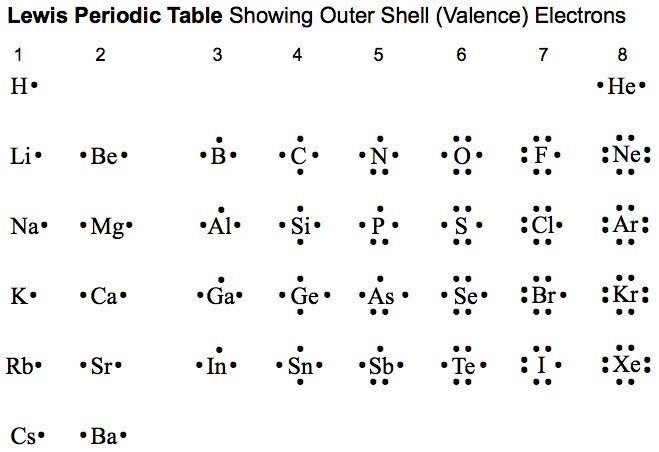
KEY IDEA: Atoms combine to form compounds. Forces of attraction between atoms establishes the chemical bond. Generally, ionic bonding and covalent bonding represent opposite extremes in a range of bond "types."
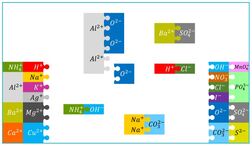
ions
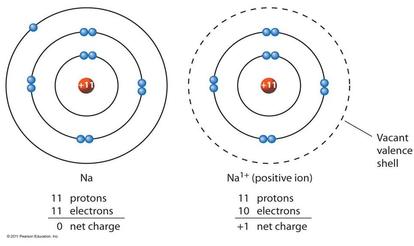
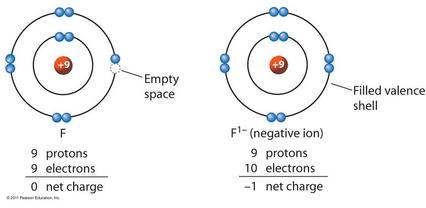
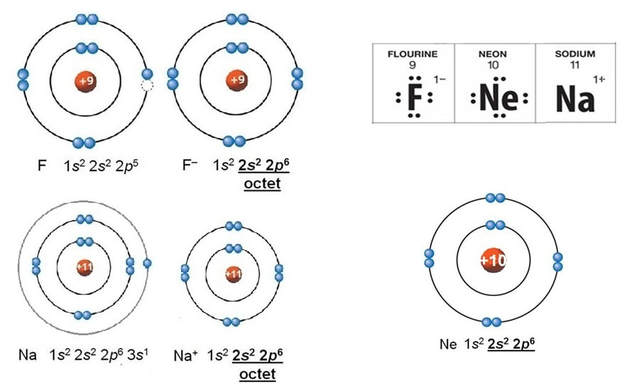
An ion is a charged atom, formed by the gain or loss of (negatively charged) electrons
- loss of electron(s) - positive ion - "cation"
- gain of electron(s) - negative ion - "anion"
An unpaired electron is called a "free radical."
Science Channel's "100 Greatest Discoveries" Electrons Form Chemical Bonds
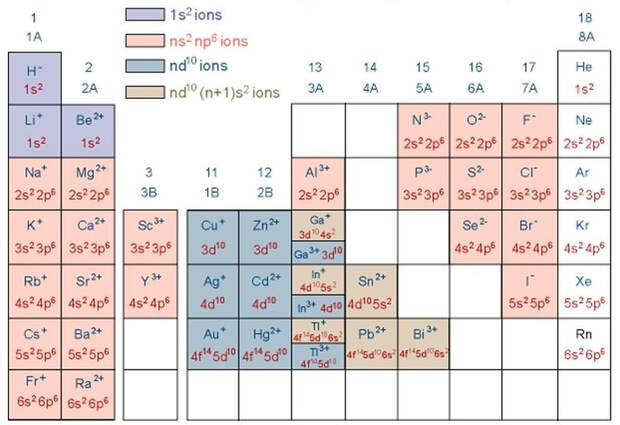
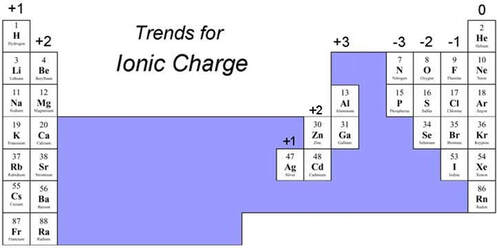
the octet rule
Ionic bonds & Ionic compounds
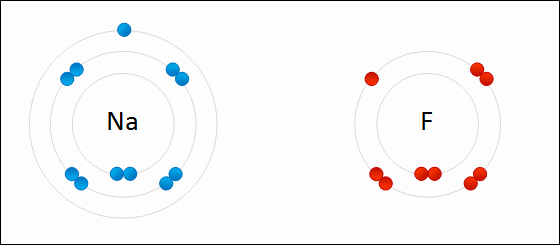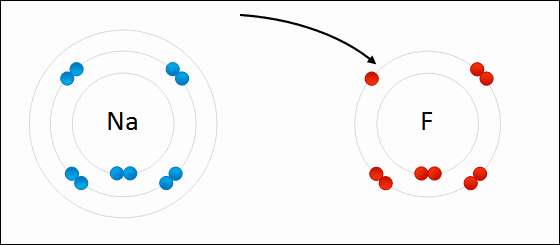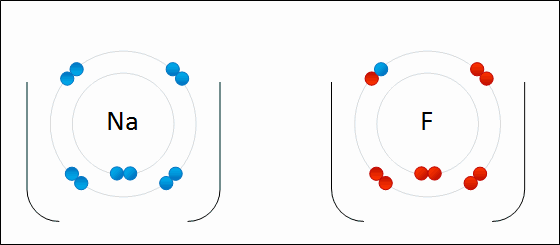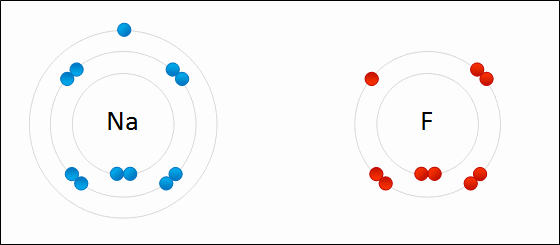
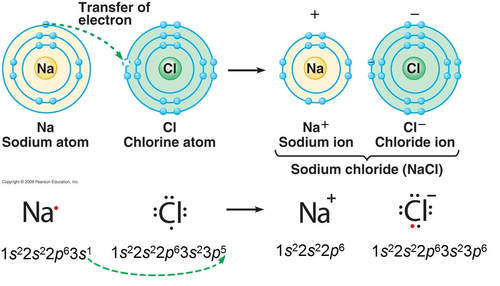
The ionic bond involves a transfer of one or more electrons from one atom (usually a metal) to another atom (usually a non-metal). The two atoms become electrically charged and are attracted by opposite charge.
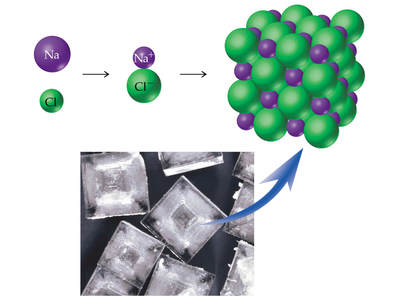
Ionic compounds are composed of cations and anions, bonded by an electrostatic attraction of opposite charge
Ionic compounds are composed of cations and anions, bonded by an electrostatic attraction of opposite charge
Formation of Ionic Bond:
Ionic Model (Whiteboard) - JavaLab
|
Reference Tables:
|
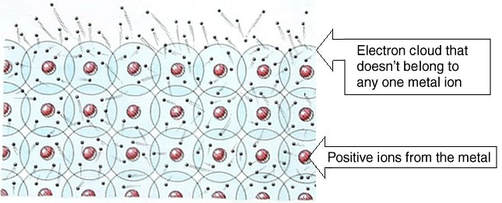
Metallic Bonding
The valence electrons of a metal can be modeled as a "sea of electrons." The valence electrons are free to move.
The metallic bond consists of the attraction of free-floating valence electrons for the positively-charged metal ions:
The metallic bond consists of the attraction of free-floating valence electrons for the positively-charged metal ions:
The "sea of electrons" model explains many of the metallic properties: conductivity, malleability, and ductility.
Copper, for example, has the highest electrical conductivity rating of all non-precious metals.
Copper, for example, has the highest electrical conductivity rating of all non-precious metals.