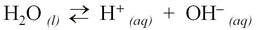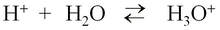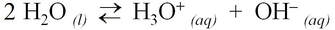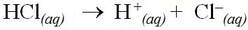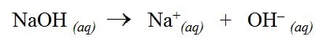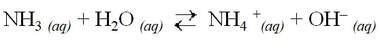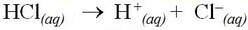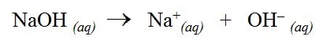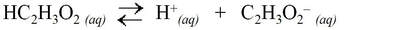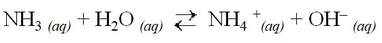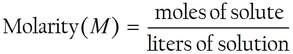KEY IDEA: Acid and bases are an important class of compounds; their reactions illustrate basic chemical principles.
Historically
Acids and bases were classically defined by physical, observable properties.
Modern definitions involve chemical behavior and molecular structure.
Self-ionization of water
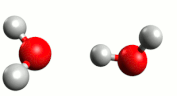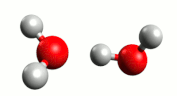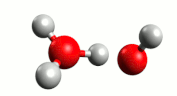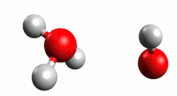
Although water is a molecule, it does self-ionize very slightly, forming hydrogen ions (H+) and hydroxide ions (OH–):
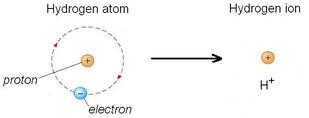
A hydrogen ion (H+) is just a proton:
In reality, a proton does not exist in an aqueous solution; it attaches to another water molecule to form the hydronium ion, H3O+.
The equation for the self-ionization of water can be written more completely:
For water, the H+ and OH– are produced in equal number. When acids and bases are dissolved in water, the number of these ions changes.
ARRHENIUS Definition
|
Examples of an Arrhenius acid include:
|
Examples of an Arrhenius base include:
|
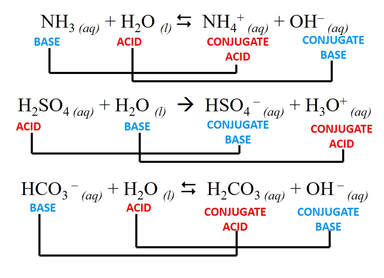
Brønsted-Lowry definition
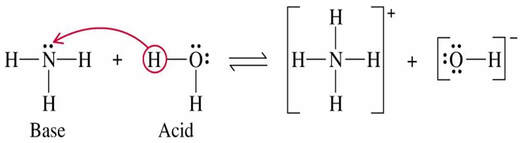
Brønsted-Lowry bases can create basic solutions by producing OH– (like a base). For example, ammonia is a common Brønsted-Lowry base.
The Brønsted-Lowry definition introduces the idea of "conjugate acid-base pairs." When an acid donates a H+, the resulting anion is now in a position to accept a proton and act as a base. Also, a base that accepts a proton can now act as an acid and donate the proton back.
Water can act as an acid or a base. Such substances are said to be amphoteric.
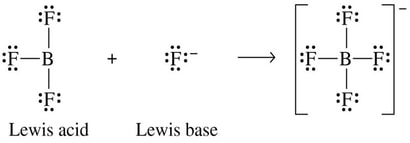
Lewis Definition
For something to act as a Lewis acid, it needs to want electrons. For example, the boron in boron trifluoride is electron-poor and short of the preferred octet with an empty orbital. BF3 can accept a pair of electrons, making it a Lewis acid:
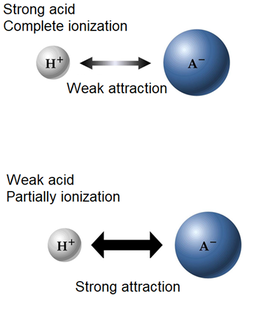
Acid-base strength
The strength of an acid or a base refers to the tendency to ionize, or the degree of ionization.
- Strong acids-bases completely ionize (100%).
- Weak acids-bases do not completely ionize; they only partially ionize.
For example, hydrochloric acid is a strong acid and completely dissociates into aqueous protons and chloride ions:
Sodium hydroxide is a strong base and completely dissociates into aqueous sodium ions and hydroxide ions:
Acetic acid, HC2H3O2, (vinegar) is a common weak acid:
Ammonia is a common weak base:
Commonly, there are 6 strong acids and 6 strong bases, all other acids-bases can be considered "weak."
Generally, the Group I and Group II metal hydroxides are considered strong bases.
Other common weak acids:
Other common weak acids:
- H2SO3 - sulfurous acid
- H3PO4 - phosphoric acid
- HNO2 - nitrous acid
- HF - hydrofluoric acid
- C6H5COOH - benzoic acid
- HC3H3O2 - acetic acid
The "concentration" of solution is defined as molarity; amount of substance (moles) per liter volume of solution.
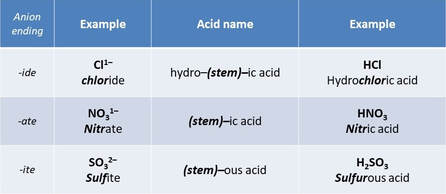
Naming acids
Acid names stem from the anion.
For simple, binary hydrogen compounds (of nonmetals when dissolved in water), the prefix "hydro- is used to represent hydrogen, followed by the name of the nonmetal, and replacing the suffix with -ic.
For simple, binary hydrogen compounds (of nonmetals when dissolved in water), the prefix "hydro- is used to represent hydrogen, followed by the name of the nonmetal, and replacing the suffix with -ic.
Naming Common Acids
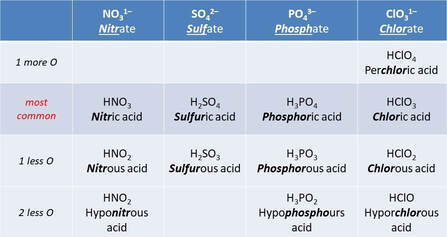
Oxoacids acids are anions contains oxygen.
Naming Oxoacids
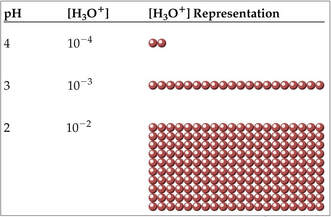
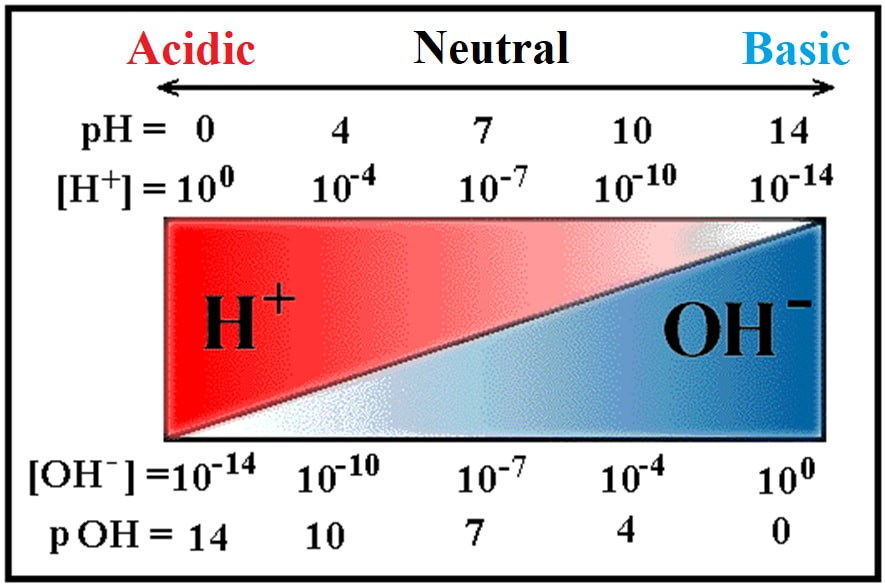
pH scale
Acid-base molarities can vary over many orders of magnitude, from 10^14 to 10^–14.
Danish chemist Søren Peter Lauritz Sørensen utilized logarithm scale (based on powers of 10) to define "pH."
In general, a pX scale is is a way to express the value of X as a logarithm.
For example, "What is log 5?" That is, what power do you raise 10 to equal 5?
Danish chemist Søren Peter Lauritz Sørensen utilized logarithm scale (based on powers of 10) to define "pH."
In general, a pX scale is is a way to express the value of X as a logarithm.
For example, "What is log 5?" That is, what power do you raise 10 to equal 5?
pH is based on the log of the hydrogen ion concentration [H+] (i.e. hydronium ion [H3O+].
For example, if one solution has a pH of 1 and a second solution has a pH of 2, the first solution is not twice as acidic as the second—it is ten times more acidic.
Equally define "pOH," based on the log of the hydroxide ion concentration [OH–].