KEY IDEA: Temperature and pressure induce changes in the state of substances.
- The physical state of matter - solid, liquid, gas - is an everyday example of the organization of subatomic particles.
- Changes in state of matter (freezing, boiling, condensation, etc.) involves a rearrangement of the particles.
Properties of Solutions
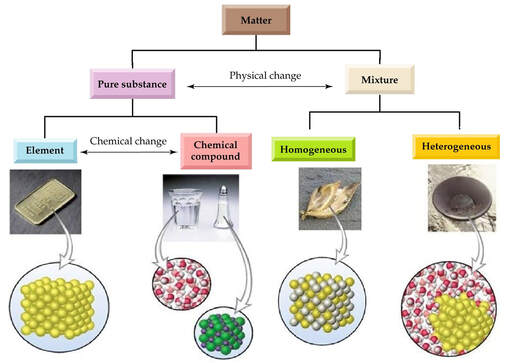
Recall... mixtures are physical blends of substances. If the mixture appears as one distinct phase, it is homogenous.
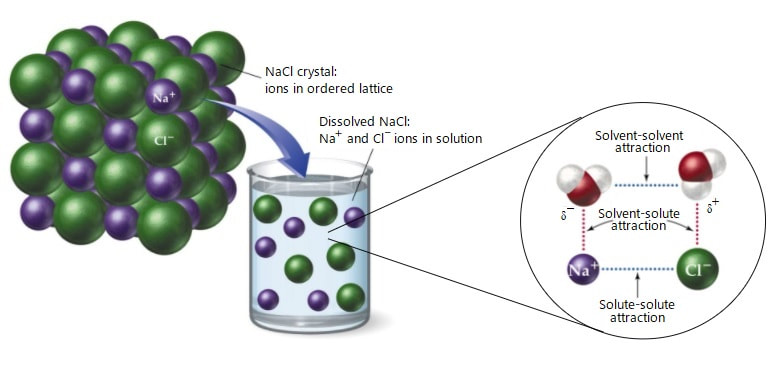
In chemistry, a solution is a homogeneous mixture composed of a solute dissolved in another substance, called the solvent. The solvent does the dissolving.
Solutions can be solid, liquid, or gas.
Solutions can be solid, liquid, or gas.
|
Solute
Gas Liquid Solid Gas Liquid Solid Gas Liquid Solid |
Solvent
Gas Gas Gas Liquid Liquid Liquid Solid Solid Solid |
Example
Atmospheric air Fog (water in air) Smog (air pollution) Carbonated water Alcohol in water Aqueous NaCl solution (saline) Hydrogen in palladium Mineral in paraffin Alloys, dental fillings |
Salt in water form electrolytes; when dissolving, salts dissociate into ions.
Glucose (sugar) readily dissolves in water, but does not dissociate into ions in solution. It is referred to as a non-electrolyte.