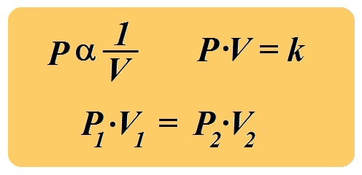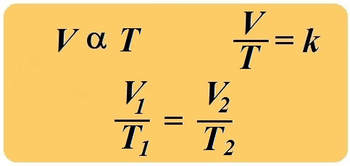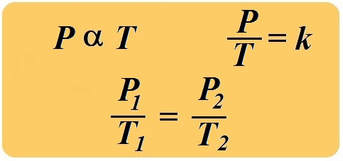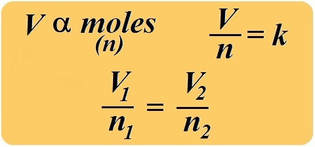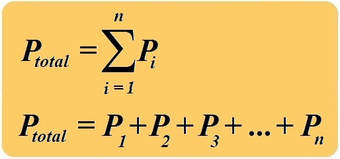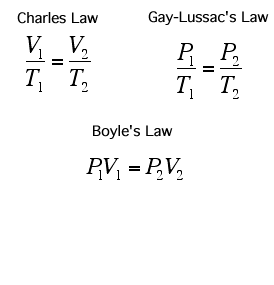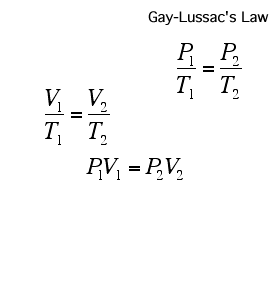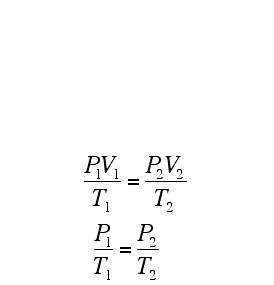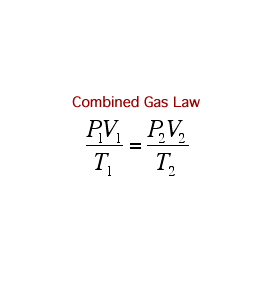|
KEY IDEA: The gaseous state of matter is the easiest to explain theoretically and therefore the easiest to understand conceptually. The assumption that gasses are considered to be made up of particles in constant, rapid motion, virtually independent of each other, forms the basis of kinetic theory. The macroscopic properties of gases reflect this microscopic behavior. |
Properties of Gases
|
The gas phase is a totally disordered state of matter in which the particles are in constant, random, rapid motion and spaced widely apart. For condensed states of matter (solids and liquids), the particles are in virtual contact and are constrained from moving freely. (The word "gas" is derived from the Greek word for chaos.)
|
The particles of a gas may be monoatomic or diatomic elements, or molecular compounds:
Although gasses vary widely in their individual chemical properties, all gases share many similar physical properties.
All gasses can be described in terms of 4 variables: (along with common units)
As different as these properties may seem, their relationships underline some of the most fundamental theories and laws of chemistry.
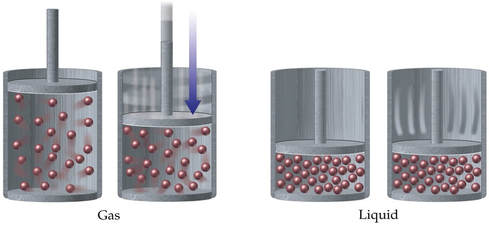
Gases differ from liquids and solids most obviously with respect to density. Even the densest gas under normal conditions is less than half as dense as the least dense solid or liquid.
Furthermore, gases are compressed more easily than liquids and solids due to the relatively large space between the particles of a gas. It is often said that gases are mostly empty space.
All gasses can be described in terms of 4 variables: (along with common units)
- Volume (liters)
- Pressure (Pascal)
- Temperature (Kelvin)
- Amount (moles)
As different as these properties may seem, their relationships underline some of the most fundamental theories and laws of chemistry.
Gases differ from liquids and solids most obviously with respect to density. Even the densest gas under normal conditions is less than half as dense as the least dense solid or liquid.
Furthermore, gases are compressed more easily than liquids and solids due to the relatively large space between the particles of a gas. It is often said that gases are mostly empty space.
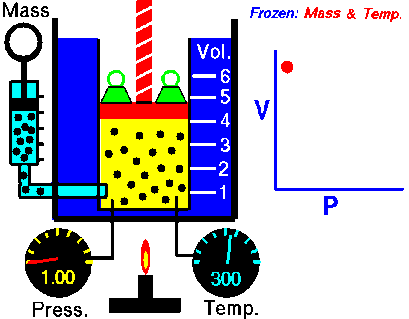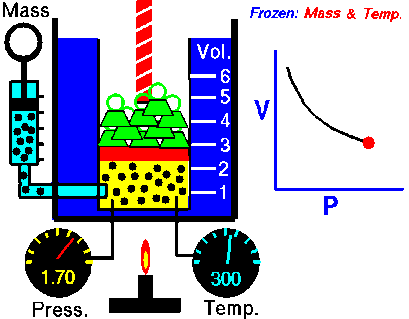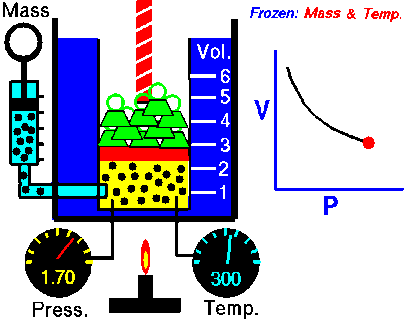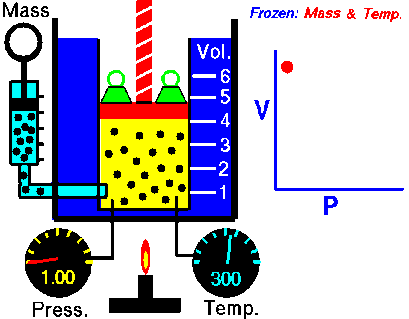
Under pressure, the gas is compressed; the particles of a gas are forced closer together. The smaller volume increases collisions between particles and with the walls of the container, which increases the pressure the gas exerts inside its container. In turn, as pressure decreases, the volume of the gas increases.
The Gas Laws
The long road to a law unifying the basic properties of gases begins in the 17th century with the Anglo‑Irish philosopher, theologian, and chemist Robert Boyle.
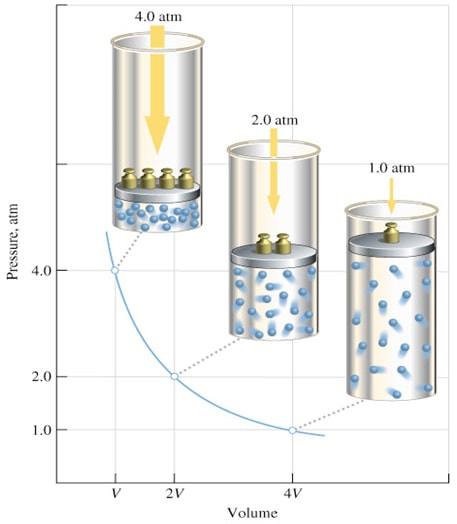
boyle's law
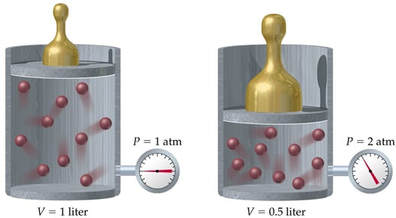
In 1662, Robert Boyle discovered that for a confined gas, the product of pressure and volume was nearly a constant; in other words, the pressure of a gas varies inversely with its volume.
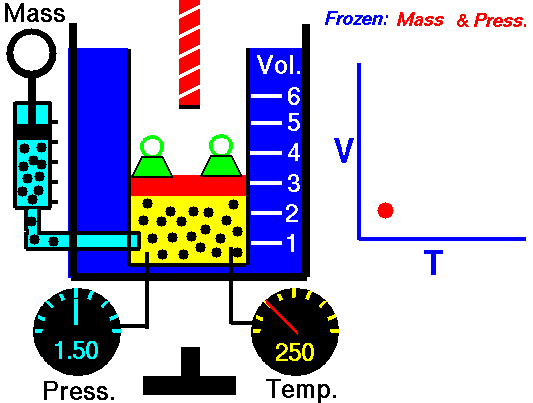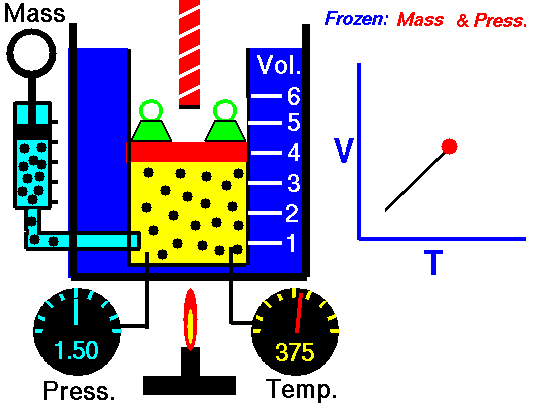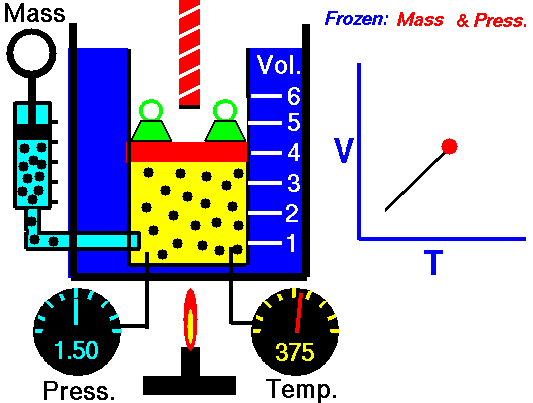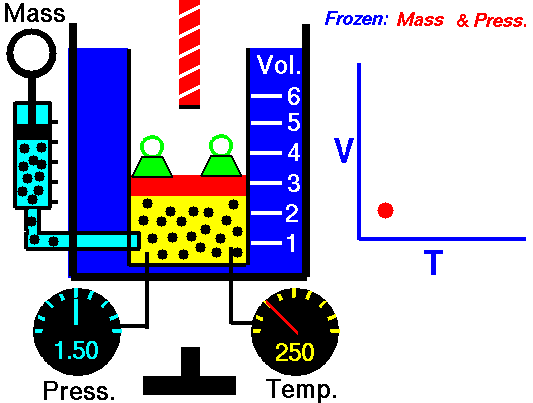
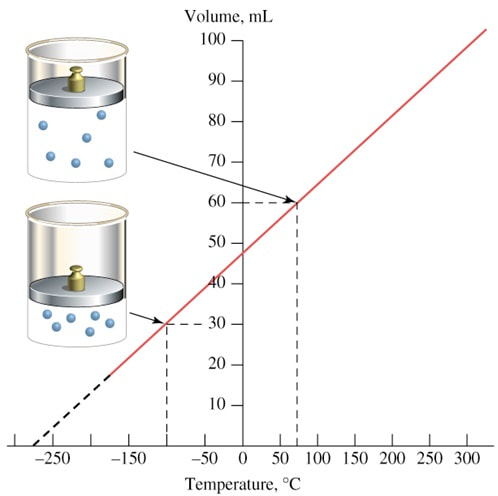
charles' law
In 1787, Jacques Charles studied the effect of temperature on the volume of a gas at constant pressure. Charles observed a linear graph of gas volume versus temperature. At constant pressure, the volume of a fixed amount of gas is directly proportional to its temperature.
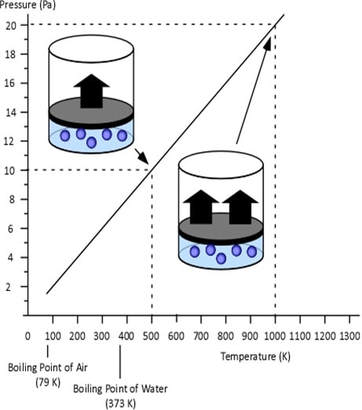
GAY-LUSSAC'S (AMONTONS') LAW
If a gas's temperature increases, so does its pressure if the mass and volume are kept constant.
This law is also referred to honor of Guillaume Amontons, who discovered the pressure-temperature relationship with atmospheric air in 1702. In 1809, Joseph Gay-Lussac experimented with different gases, and attributed his findings to much of Charles's unpublished data.
This law is also referred to honor of Guillaume Amontons, who discovered the pressure-temperature relationship with atmospheric air in 1702. In 1809, Joseph Gay-Lussac experimented with different gases, and attributed his findings to much of Charles's unpublished data.
avogadro's Law
Equal volumes of all gases at the same temperature and pressure contain the same number of particles.
One mole of a gas occupies 22.4 L, at STP (standard temperature and pressure). This is known as MOLAR VOLUME.
One mole of a gas occupies 22.4 L, at STP (standard temperature and pressure). This is known as MOLAR VOLUME.
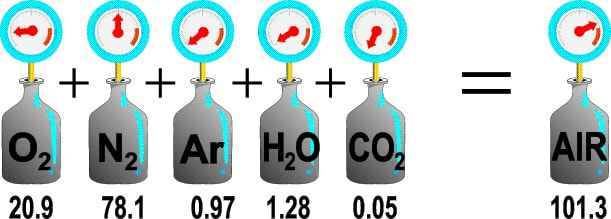
DALTON'S LAW OF PARTIAL PRESSURES
The total pressure of a mixture of gases is equal to the sum of partial pressures of each individual gas.
LINK: Online investigations for the gas laws (University of Oregon Chemistry Dept.)
Additional Resources
LINK: Animated Gas Laws (hosted by NASA)
LINK: Animated Gas Laws (hosted by NASA)
COMBINED GAS LAW
The relationships between pressure, volume, and temperature for a fixed amount of gas can be used to determine a set of conditions, given an initial set of conditions:
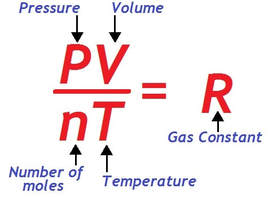
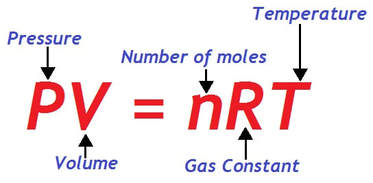
ideal gas law
The combined gas law can be accommodated to include the variable "moles" to calculate the amount of a confined gas. If we use Avogadro’s law, which states the amount, or moles of gas (n) is proportional to its volume, we obtain the expression:
The universal gas constant, denoted "R", is a constant of proportionality. The value of R depends upon the units of pressure and volume.
"Ideal" gases
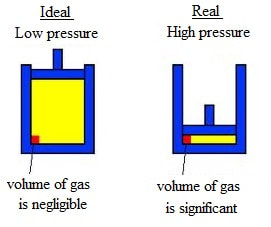
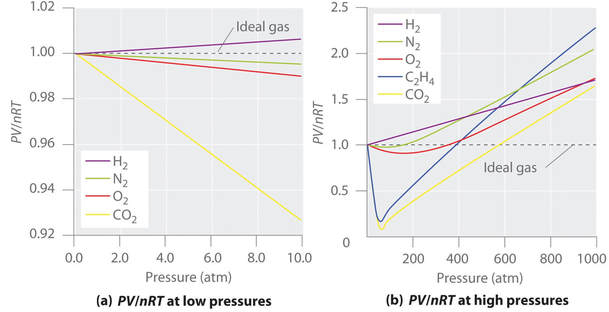
An ideal gas follows the gas laws at ALL pressures and temperatures, thus conforming precisely to kinetic theory. Kinetic theory ignores the volume of the gas particles themselves and all interactions between the particles. Under extreme pressure and temperature conditions, these two assumptions (negligible volume and negligible interactions) are no longer valid. Since kinetic theory assumes gas particles to have zero volume, the volume available to them for motion is the same as the volume of the container. However, molecules have small yet measurable volume.
At low pressures, gas molecules are spaced far apart. As pressure of the gas increases, the intermolecular distances between molecules decreases, and the volume occupied by the molecules becomes significant compared with the volume of the container. As a result, the total volume occupied by the gas is greater than the volume predicted by the ideal gas law.
All particles interact to some extent with one another due to a combination of forces. Intermolecular forces are particularly significant at low temperatures and high pressures, where intermolecular distances are shorter. Forces of attraction between molecules reduces the pressure exerted by the gas on the container wall decreases, thus the observed pressure is less than expected.
All particles interact to some extent with one another due to a combination of forces. Intermolecular forces are particularly significant at low temperatures and high pressures, where intermolecular distances are shorter. Forces of attraction between molecules reduces the pressure exerted by the gas on the container wall decreases, thus the observed pressure is less than expected.
For an ideal gas, a graph of PV/nRT vs. pressure yields a horizontal line with a y-intercept equal to 1 (see below). Real gases deviate significantly from ideal behavior, particularly at very low pressure (a) and very high pressure (b):
|
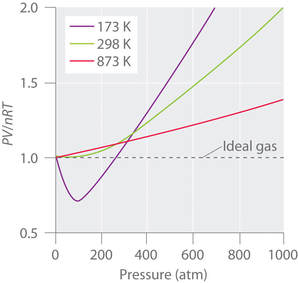
Real gases also deviate from expected ideal gas behavior at very high temperature. A plot of PV/nRT vs. pressure for nitrogen gas at different temperatures shows the deviation from ideal behavior as the temperature increases.
At high temperatures, the molecules have sufficient kinetic energy to overcome intermolecular attractive forces and the effects of nonzero molecular volume predominate. Conversely, as the temperature is lowered, the kinetic energy of the gas molecules decreases. Eventually, a point is reached where the molecules can no longer overcome the intermolecular attractive forces, and the gas condenses to a liquid. At low temperatures, the ratio of PV/nRT is lower than predicted for an ideal gas. |
VIDEO: Gas Law Demonstrations