KEY IDEA: Covalent bonding (the opposite extreme of ionic bonding) involves the sharing of one or more pairs of electrons. A theory of bonding helps rationalize the shapes of molecules.
Nature of covalent bond
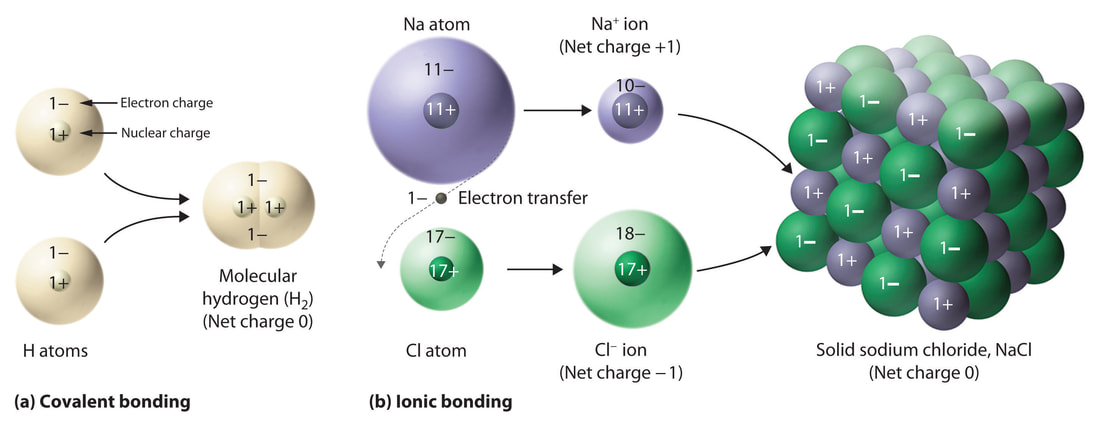
Covalent bonding is the result of two atoms SHARING one or more electrons to complete their valence.
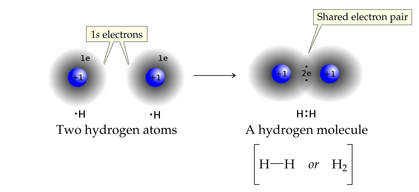
The simplest element is hydrogen. An atom of hydrogen has one electron, but needs two electrons to complete its valence.
The simplest element is hydrogen. An atom of hydrogen has one electron, but needs two electrons to complete its valence.
Two hydrogen atoms can share a pair of electrons, and complete the valence orbital for each.
This is the basis for a SINGLE covalent bond, a sharing of TWO electrons.
The structural formula, H:H, represents the shared pair of electrons of the covalent bond as two dots ":"
Hydrogen is unique, in that 2 electrons complete the valence. Most elements seek 8 electrons in their valence.
This is the basis for a SINGLE covalent bond, a sharing of TWO electrons.
The structural formula, H:H, represents the shared pair of electrons of the covalent bond as two dots ":"
Hydrogen is unique, in that 2 electrons complete the valence. Most elements seek 8 electrons in their valence.
GROUP 17 (7A)
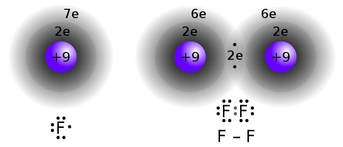
In the case of the halogens, such as fluorine, an atom has 7 valence electrons, 1 electron short of a complete valence:
In the case of the halogens, such as fluorine, an atom has 7 valence electrons, 1 electron short of a complete valence:
Two atoms of fluorine can each donate 1 electron to form a shared pair, and both atoms achieve a completed valence.
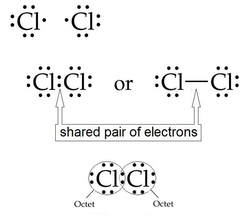
All the halogens form SINGLE covalent bonds. Similarly, the electron-dot structure for chlorine:
All the halogens form SINGLE covalent bonds. Similarly, the electron-dot structure for chlorine:
GROUP 16 (6A)
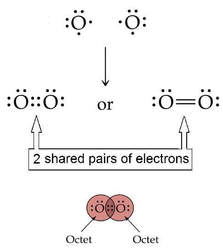
Oxygen can share multiple pairs of electrons to complete valences:
Oxygen can share multiple pairs of electrons to complete valences:
Atoms of oxygen share TWO PAIRS of electrons, thus forming a DOUBLE covalent bond.
GROUP 15 (5A)
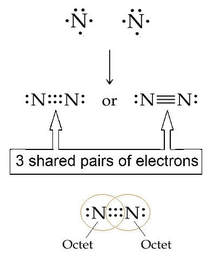
Two atoms of nitrogen can share 3 pairs of electrons to complete their valence:
Two atoms of nitrogen can share 3 pairs of electrons to complete their valence:
Atoms of nitrogen share THREE PAIRS of electrons, thus forming a TRIPLE covalent bond
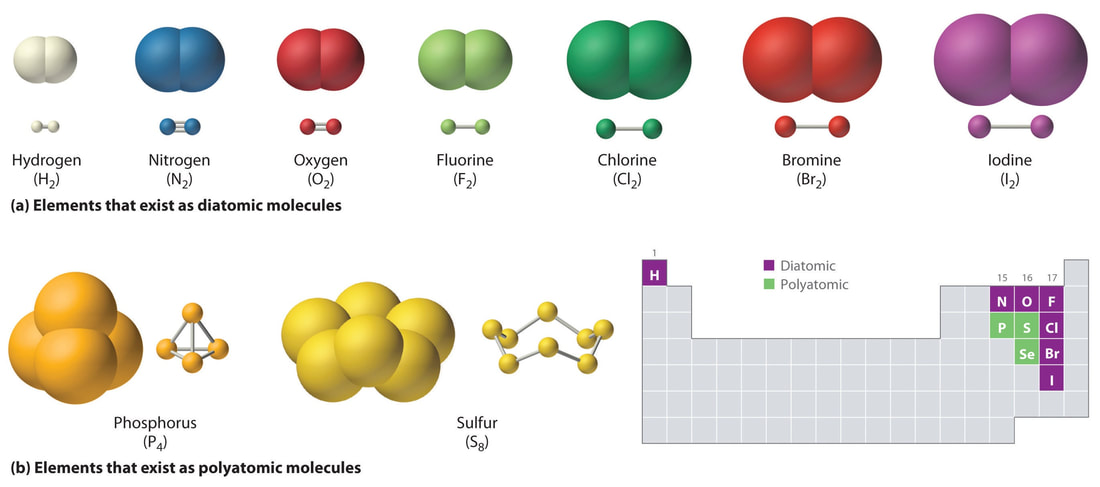
MOLECULAR ELEMENTS
MOLECULAR COMPOUNDS
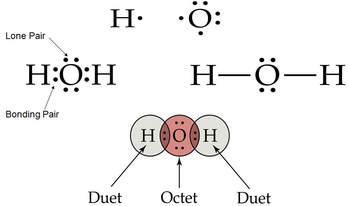
A molecule of water consists of 2 hydrogen atoms and 1 oxygen atom.
The electron-dot structure for a water molecule:
The electron-dot structure for a water molecule:
A pair of valence electrons that is not shared between atoms is an unshared pair (a.k.a. lone pair, or non-bonding pair).
COVALENT Vs. IONIC BONDING
BOND STRENGTH
or "Bond Dissociation Energy"
- Energy required to break a covalent bond, Units expressed per mole
- LARGE value means STRONG covalent bond
- Multiple bonds increase bond strength
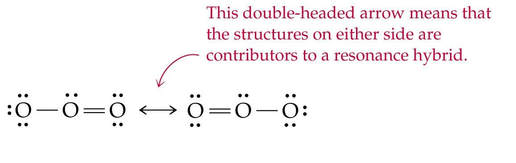
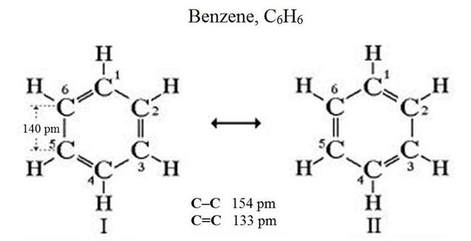
resonance
- the delocalization of an electron pair over several atoms.
- spreads the double-bond character evenly over the atoms.
Molecules are said to "resonate" between different arrangements. Both arrangements exist in any sample of that substance.
Benzene is a carcinogen. The molecular structure satisfies the octet rule by arranging carbon atoms in a ring.
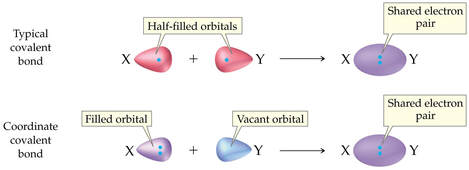
COORDINATE COVALENT BOND
- When 1 atom donates both pairs of electrons to form the shared pair.
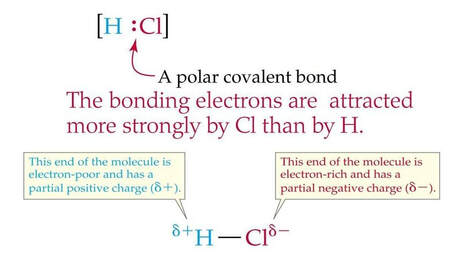
Polar bonds and molecules
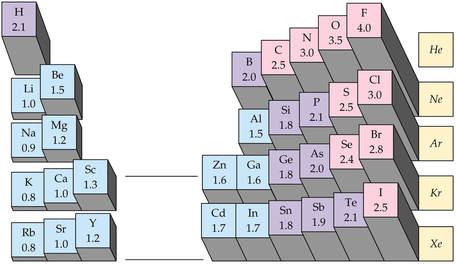
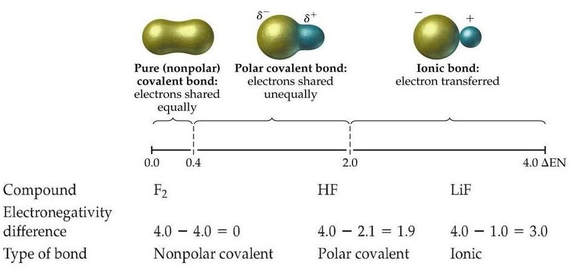
Recall electronegativity is defined as an atom's ability to attract shared electrons in a bond.
The difference in electronegativity determines bond polarity:
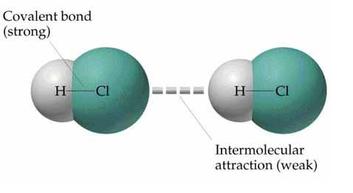
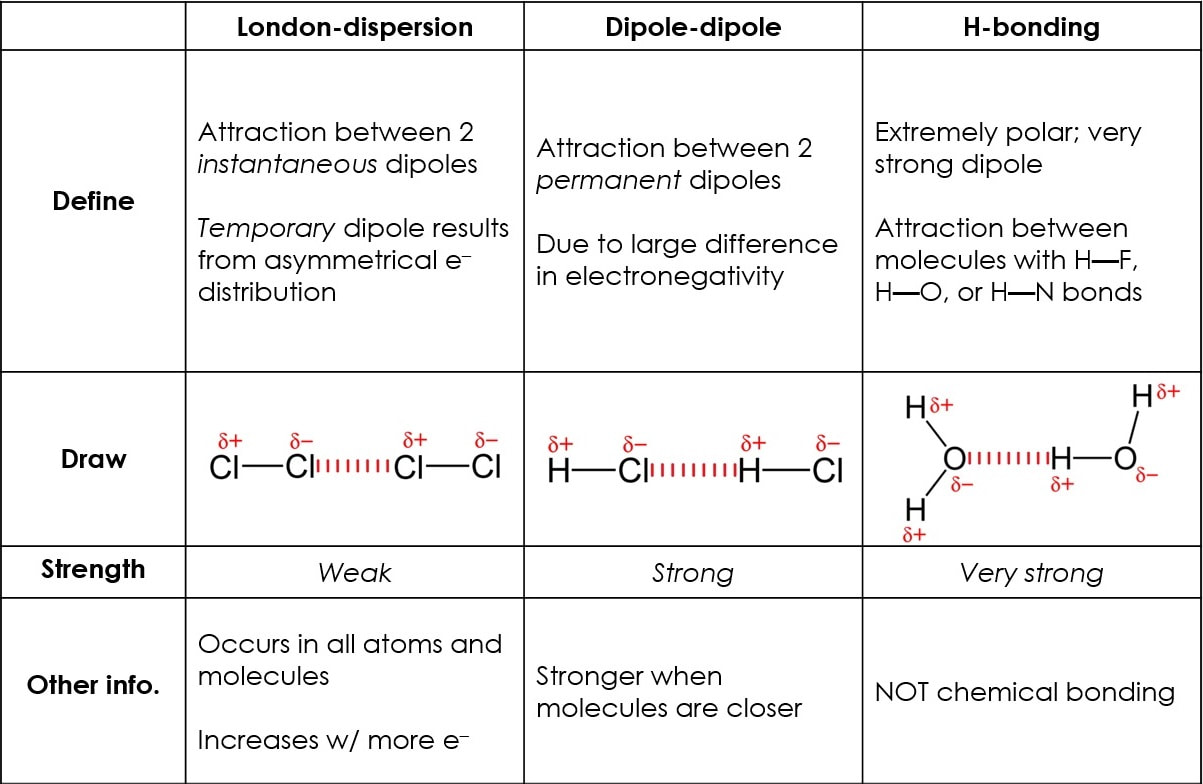
Intermolecular forces of attraction ("I.M.F.")
The forces that act between molecules play a significant role in many aspects of chemistry. The boiling point of liquids and the solubility of solids, even the structure of DNA and proteins, all depend on intermolecular forces.
Intermolecular forces are summarized on the following table:
MOLECULAR GEOMETRY
Valence Shell Electron Repulsion Theory (VSEPR) - "ves per"