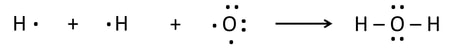KEY IDEA: Change is a trademark of chemistry. Atoms are rearranged in a chemical reaction to form new materials. All chemical reactions involve the rearrangement of electrons through the breaking and reforming of chemical bonds.
Chemical Reactions
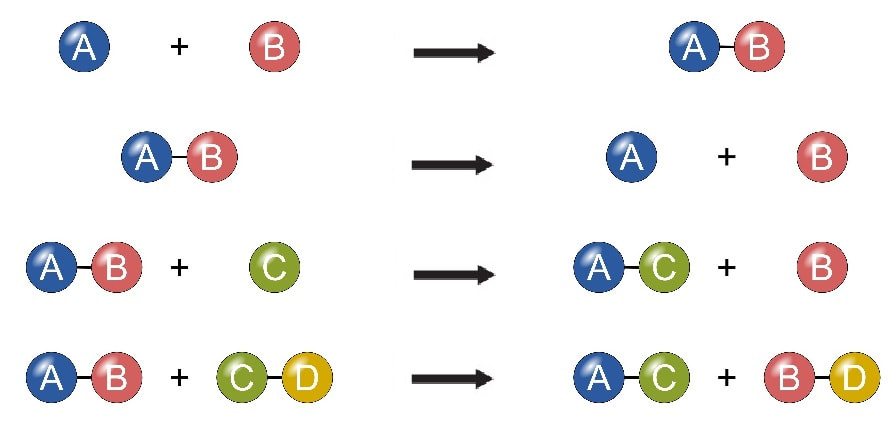
Chemical reactions are processes where one or more substances are transformed into one or more different substances.
Chemical equations are a shorthand notation describing a chemical reaction between substance.
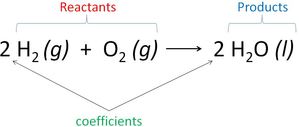
The general form of an equation: Reactants → Products.
A reactant is the starting material of a reaction; the substances undergoing reaction. The product is the material produced in the reaction.
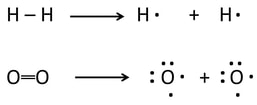
Bonds are broken in the reactants, atoms are rearranged, and new bonds are formed. This is the process of any reaction.
A chemical equation must be consistent with "chemical facts." This means that all reactants and products observed in a reaction must be included in the equation.
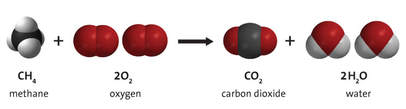
Example Hydrogen and oxygen react to form water:
Chemical equations are a shorthand notation describing a chemical reaction between substance.
The general form of an equation: Reactants → Products.
A reactant is the starting material of a reaction; the substances undergoing reaction. The product is the material produced in the reaction.
Bonds are broken in the reactants, atoms are rearranged, and new bonds are formed. This is the process of any reaction.
A chemical equation must be consistent with "chemical facts." This means that all reactants and products observed in a reaction must be included in the equation.
Example Hydrogen and oxygen react to form water:
- Bonds are broken in the reactants:
- Atoms are rearranged, and new bonds are formed:
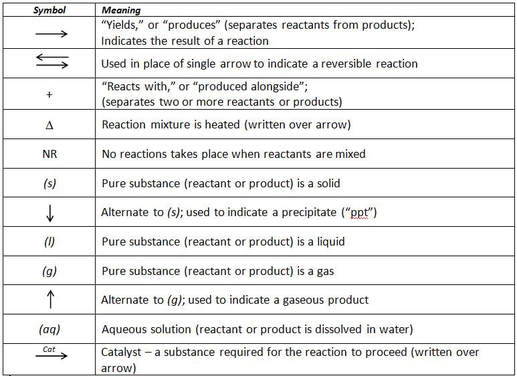
The following table summarizes the common symbols used to describe chemical reactions and their meaning:
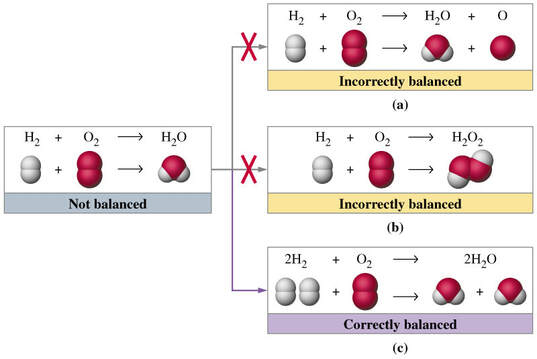
A chemical equation follows the Law of Conservation of Mass. Atoms are neither created nor destroyed in chemical reactions. Coefficients are used to balance the numbers of atoms. An unbalanced equation is called a "skeleton equation."
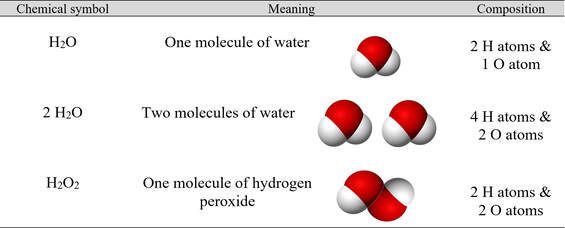
Note the difference between a coefficient and a subscript:
VIDEO: An introduction to balancing chemical equations.
Here are a few general tips for balancing equations:
Here are a few general tips for balancing equations:
- Do not balance equations in your head.
- Never begin balancing elements that occur in more than one compound.
- Balance the elements in the more "complex" compounds first.
- Simplify the process by balancing polyatomic ions as whole units.
- Balance "free" elements last.
- If necessary, balance with fractions. Clear or simplify fractions by multiplying or dividing through the equation.
- When the equation is correctly balanced, the coefficients should be in lowest terms.
- If elements do not balance, start over.
PhET hosts an online simulation for balancing equations:
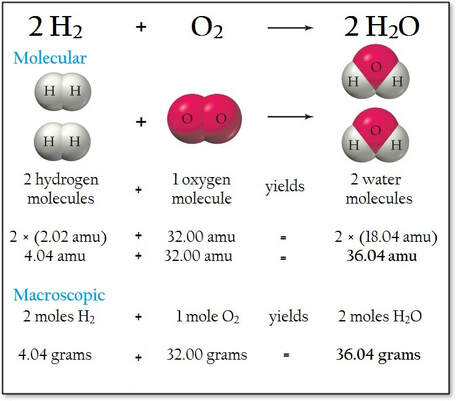
A balanced equation provides quantitative relationships among the reactants and products in a chemical reaction:
Note, only atoms and mass are conserved. This is true for all chemical reactions.
A balanced equation can be used to calculate the necessary weight relationships in the reaction.
A balanced equation can be used to calculate the necessary weight relationships in the reaction.
|
Chemical reactions often involve the absorption or emission of energy, usually in the form of heat.
The formation of water from elemental hydrogen and oxygen is an example of an exothermic reaction: energy is released. Conversely, an endothermic reaction is one that absorbs energy. LINK What Really Felled the Hindenburg |
Classifying Reactions
Many chemical reactions can be categorized as one of 7 general "types":
Many chemical reactions can be categorized as one of 7 general "types":
|
1. Synthesis
Two reactants combine to form 1 product. a.k.a. called "Combination" 2. Decomposition A single compound breaks down into simpler products. 3. Single-Replacement An element reacts with a compound and displaces an element in the compound. 4. Double-Replacement Two ionic compounds exchange ions and form two new compounds |
5. Acid-Base
A double-replacement involving an acid and a base; product is a salt and water.
6. Combustion is a reaction with oxygen gas. Typically gives off LOTS of energy (light and heat).
General form: A + O2 → AO (product is an oxide)
A double-replacement involving an acid and a base; product is a salt and water.
- Example: Acetic acid (vinegar) and sodium bicarbonate (baking soda, a base.)
6. Combustion is a reaction with oxygen gas. Typically gives off LOTS of energy (light and heat).
General form: A + O2 → AO (product is an oxide)
- Combustion of a hydrocarbon (any compound containing C and H) produces carbon dioxide and water.
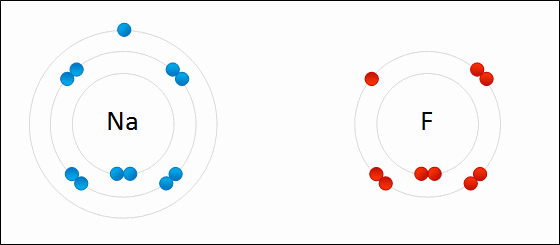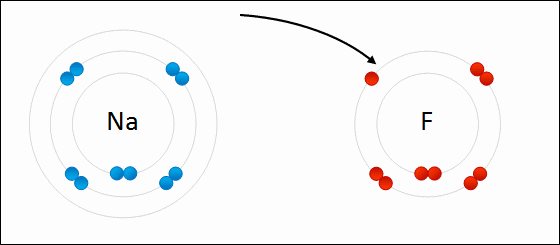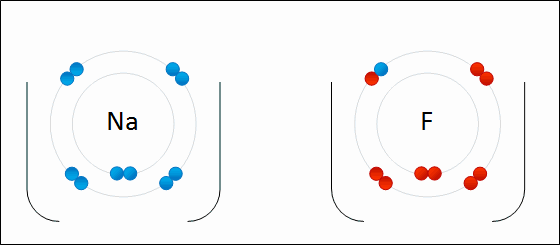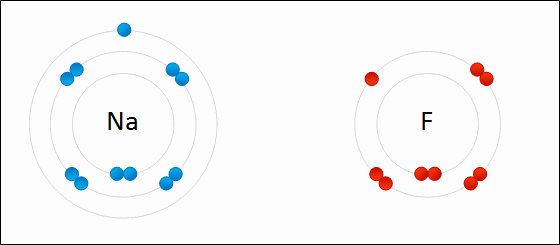
7. Reduction-Oxidation (or "Redox")
A reaction involving the transfer of electrons:
A reaction involving the transfer of electrons:
- Oxidation is the loss of electrons; increases in oxidation number.
- Reduction is the gain of electrons; decrease in oxidation number
YouTube Awesome Chemical Reactions!
Ted-Ed. The Haber Process
Ted-Ed. How to speed up reactions (and get a date)
Ted-Ed. The Haber Process
Ted-Ed. How to speed up reactions (and get a date)