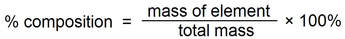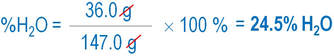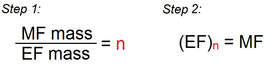KEY IDEA: The mole is (simply) a number. The unit provides chemists with a way to "count" atoms and molecules by weighing them.
The Mole
One of the most important concepts in an introductory chemistry course is the mole. The mole is analogous to a dozen: a word that means a quantity, or a number.
The mole is the unit for the amount of something. (As in "how many," or "how much.") Conceptually, the mole is a way of counting things, like atoms, and molecules.
In 1811, Italian chemist Amedo Avogadro proposed that a volume of gas is proportional to the number of particles.
The mole is the unit for the amount of something. (As in "how many," or "how much.") Conceptually, the mole is a way of counting things, like atoms, and molecules.
In 1811, Italian chemist Amedo Avogadro proposed that a volume of gas is proportional to the number of particles.
Avogadro's Law
One mole of something equals 6.022 × 10^23 "particles" of that something.
The quantity 6.022 × 10^23 is set as the number of carbon-12 atoms in 12.0 grams.
The mole is also named "Avogadro's Number" in his honor.
- One mole of C is 6.022 × 10^23 carbon atoms
- One mole of H2O is 6.022 × 10^23 water molecules
- One mole of H2O contains one mole of oxygen (6.022 × 10^23 O-atoms) and 2 moles of hydrogen (1.204 × 10^24 H-atoms)
The quantity 6.022 × 10^23 is set as the number of carbon-12 atoms in 12.0 grams.
- 1 mole of carbon-12 atoms have a mass equal to exactly 12 grams
The mole is also named "Avogadro's Number" in his honor.
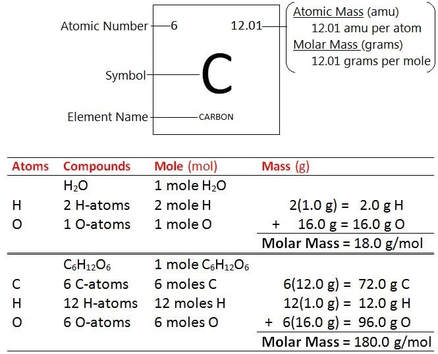
MOLAR MASS
(or gram-formula mass) is the mass (in grams) of one mole. (units: g/mole)
An atom of each element has a characteristic mass. Recall, the atomic mass for the elements (found on a periodic table) is a weighted average of the masses of all natural isotopes for that element. Atomic masses are based on the the carbon-12 isotope having a mass defined as 12.000 atomic mass units (amu). One amu = 1.66053 x 10^-24 g.
The atomic mass expressed in units of grams is the mass of one mole (6.022 × 10^23 atoms) of that element.
An atom of each element has a characteristic mass. Recall, the atomic mass for the elements (found on a periodic table) is a weighted average of the masses of all natural isotopes for that element. Atomic masses are based on the the carbon-12 isotope having a mass defined as 12.000 atomic mass units (amu). One amu = 1.66053 x 10^-24 g.
The atomic mass expressed in units of grams is the mass of one mole (6.022 × 10^23 atoms) of that element.
Online molar mass calculator.
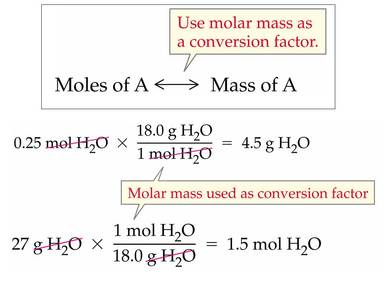
Molar mass can be used to convert between mass and moles. (Recall dimensional analysis from ch. 3.)
Molar mass can be used to convert between mass and moles. (Recall dimensional analysis from ch. 3.)
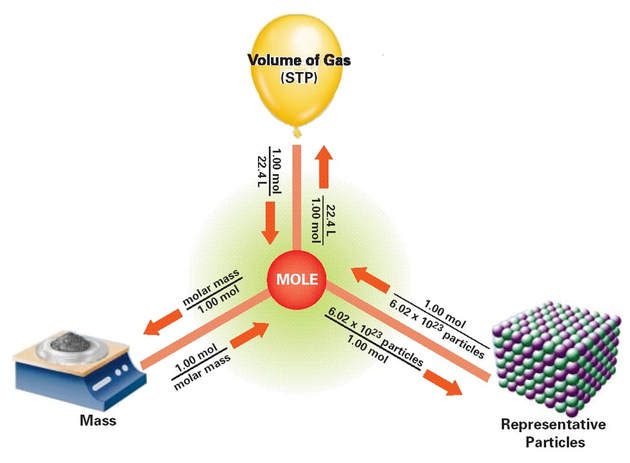
Similarly, molar volume (1 mole = 22.4 L) can be used to convert between volume and moles.
The mole is a convenient way to count items (or atoms) by weighing them or by measuring their volume.
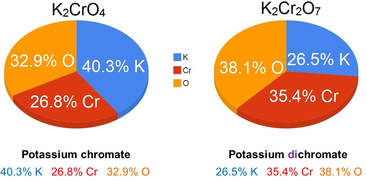
Percent composition
Percent composition is the percent by mass of each element in a compound
Mass spectrometry is a vital tool in chemical analysis The video shows how it works and covers a couple simple related exercises.
Sample calculations:
- Find the percent composition of copper(I) sulfide, Cu2S.
- How many grams of copper can be obtained from a 38.0 g sample of Cu2S?
- From the previous example, Cu2S is 80.% copper.
- Find the mass percent of water in calcium chloride dihydrate, CaCl2 • 2H2O
Empirical & Molecular Formulas
- Empirical formula is the smallest whole-number ratio of atoms in a compound
- Molecular formula is the "actual" number of atoms in a compound. (a.k.a. the "true" formula)
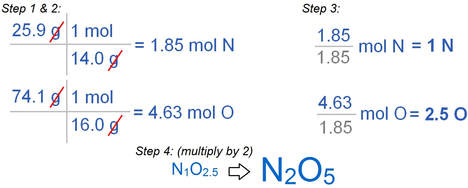
Empirical formulas can be determined from percent composition data:
- Assume 100. gram sample (unless given).
- Covert grams of each element to moles.
- Divide by smallest number of moles to find subscripts.
- If necessary, multiply by 2, 3, or 4 to get whole-numbers.
Sample calculation:
- Determine the empirical formula for a compound that is 25% nitrogen and 75% oxygen.
If the molar mass is given, the "true" molecular formula can be determined:
- Divide the molar mass by the empirical mass.
- Multiply each subscript in the empirical formula by the answer from #1.
Sample calculation:
- The empirical formula for ethylene is CH2. Find the molecular formula if the molar mass of ethylene is 28.1 g/mol.
- The empirical mass (CH2) is 14.0 g/mol.