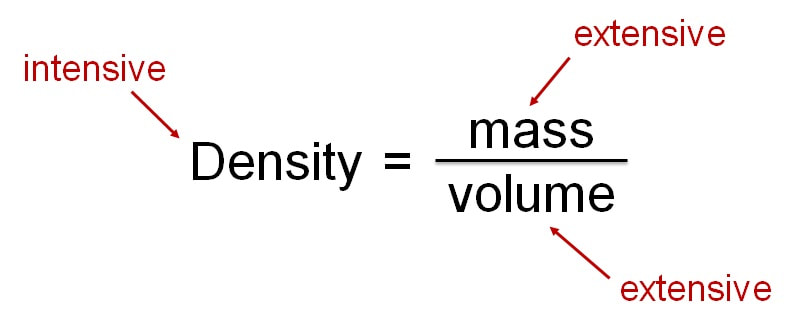KEY IDEA: Matter is the physical "stuff" of the universe. The infinite variety of matter is the result of about 100 basic building blocks.
Properties of Matter
Chemistry is the study of matter and its changes.
Understanding matter begins with observing the properties of matter: qualities or characteristics that allow you to identify matter and distinguish different forms of matter.
Properties of matter can be physical or chemical:
Understanding matter begins with observing the properties of matter: qualities or characteristics that allow you to identify matter and distinguish different forms of matter.
Properties of matter can be physical or chemical:
- Physical properties can be observed without changing the identify, or composition, of matter.
- usually the most obvious properties
- observed with the senses
- e.g. density, melting point, boiling point
- Chemical properties can ONLY be observed by changing the identity of matter.
- refers to the "behavior" of matter (as in a chemical reaction)
- matter's ability to react and produce a different form of matter.
- e.g. flammability, oxidation
Physical properties can either be extensive or intensive:
Physical properties can either be extensive or intensive:
States of matter
The state, or phase of matter (solid, liquid, gas) is a common physical property.
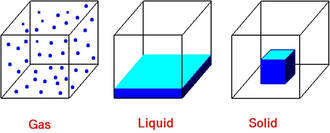
The state of matter depends upon the organization of submicroscopic particles:
The state of matter depends upon the organization of submicroscopic particles:
The physical states of matter are described in terms of shape and volume:
- Gases have indefinite shape and volume.
- Liquids have indefinite shape but definite volume.
- Solids have definite shape and volume.
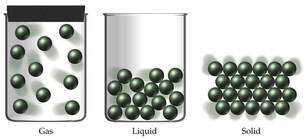
The states of matter differ from each other in the kinetic energy of the particles.
- Gas particles have very high kinetic energy; particles of a solid have very low kinetic energy:
- Gas particles are spaced very far apart from each other, and spread out to occupy all available space.
- Particles of a solid are tightly packed, arranged in an orderly pattern, and vibrate in a fixed location.
- Particles of a liquid are close together, disorderly arranged, and able to flow past one another (fluidity).
Water is commonly found in 3 states.
Explore the composition of air and learn about the different atoms and molecules that we breathe every day.
changes in matter
A physical change does not change the composition of matter: the particles are only rearranged.
The 3 states of matter can be interchanged by the addition or removal of heat:
The 3 states of matter can be interchanged by the addition or removal of heat:
Most physical changes involve a change in temperature, but changes in state can also result from a change in pressure.
Modeling the submicroscopic world
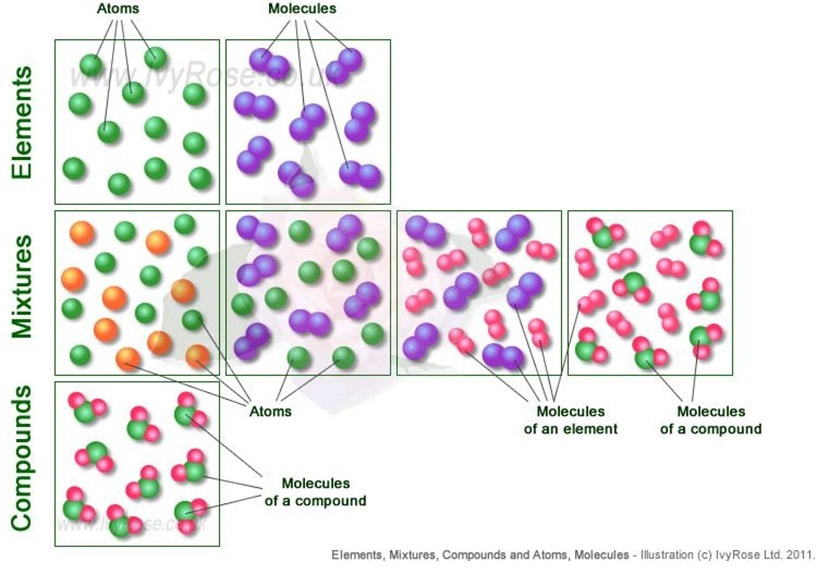
Atomic windows are graphical models to represent the submicroscopic particles. Atoms are depicted as spheres, each element of a different color. Combinations of atoms, called "molecules," depict compounds and some elements..
Molecules are a combination of atoms.
- Atoms are represented as single spheres. Spheres of the same color and size represent atoms of the same element.
- Molecules are represented by two or more spheres joined together.
- Molecules of elements are represented by two or more spheres of the same color joined together.
- Molecules of compounds are represented by two or more spheres of different color and size joined together.
chemical changes
Chemical changes occur ONLY when the there is a change in composition
- an ability to undergo a chemical change; “reactivity”
- e.g., burn, rot, decompose, ferment, explode, corrode, rust, oxidize, etc…
- can be used to identify matter
- indicated with an arrow in a chemical equation
Possible “clues” to a chemical change:
NOTE: Clues do not necessarily confirm chemical change.
- Transfer of energy
- Heat &/or light given off
- Energy (heat/electricity) is absorbed
- Change in color
- Production of gas
- Formation of precipitate (a solid that forms out of a liquid)
NOTE: Clues do not necessarily confirm chemical change.
Explore a campsite in this simulation to observe different chemical changes and physical changes:
Physical and chemical changes may be reversible or irreversible:
- Reversible: the change can “undone”; substance returns to original state
- e.g. photosynthesis, respiration, change in state
- Irreversible: the change cannot be undone; unable to retrieve original sample.
- e.g. shredding paper, burning, rusting of iron
Distinguishing substances vs mixtures
 The periodic table of elements
The periodic table of elements
SUBSTANCE– uniform & definite composition; pure
Substances are classified as elements or compounds:
LINK Explore the Elements (Royal Society of Chemistry)
MIXTURE– a physical blend of 2 or more substances.
Phase – a distinct part, or region, of a mixture.
Two types of mixtures:
- Every sample of a substance has identical intensive properties.
Substances are classified as elements or compounds:
- Element – simplest form of matter that has a unique set of properties.
- An atom is the smallest unit of an element that has all the properties of that element.
LINK Explore the Elements (Royal Society of Chemistry)
- Compound – a substance composed of 2+ elements chemically combined in fixed proportion.
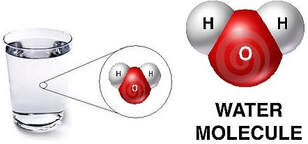
- Examples: water = H2O; table salt = NaCl; sucrose = C12H22O11
- A molecule is the smallest unit of a compound.
MIXTURE– a physical blend of 2 or more substances.
- e.g. atmospheric air = 78% nitrogen, 21% oxygen, trace carbon dioxide and argon.
Phase – a distinct part, or region, of a mixture.
Two types of mixtures:
- Heterogeneous – a mixture that is not uniform in its composition; 2+ phases
- Homogeneous – a mixture with uniform composition throughout; single phase
- Homogeneous liquids are called solutions.
Separating Mixtures
Physical properties can be used to separate mixtures. (See image below)
Physical properties can be used to separate mixtures. (See image below)
- Filtration – separating a solid from a liquid in a heterogeneous mixture; based on solubility. (a)
- Distillation – separating the components of a liquid homogeneous mixture by their boiling points. (b)
- Chromatography – separating a mixture based on a differences in solubility. (c and d)
properties of compounds
A substance has fixed composition; the composition of a mixture varies.
Compounds have different properties than the elements that make them up.
For example, iron and sulfur react to form iron sulfide:
Compounds have different properties than the elements that make them up.
For example, iron and sulfur react to form iron sulfide:
Another example: sodium and chlorine combine to form sodium chloride, common table salt.
Compounds can only be simplified by chemical properties. Compounds cannot be separated physically.
There is no chemical process that will break down elements into simpler substances.
Symbols and Formulas:
Chemists use chemical symbols to represent elements and chemical formulas to represent compounds.
Chemists use chemical symbols to represent elements and chemical formulas to represent compounds.
|
Symbol:
hydrogen = H sodium = Na chlorine = Cl oxygen = O |
Formula:
salt = NaCl water = H2O |
NOTE:
- The first letter of the symbol is always capitalized; second letter is lower case.
- Subscripts indicate the relative proportion of the elements in the compound.
- Example: Water, H2O: 2 hydrogen atoms for every 1 oxygen atom
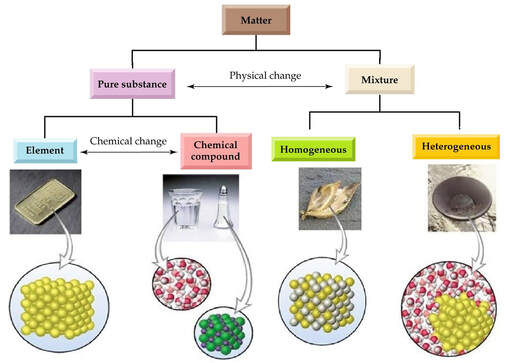
The classification of matter based on composition is summarized according to the following flow chart:
In Summary:
- Matter is classified as substances or mixtures.
- Pure substances have invariable properties and composition. Mixtures have variable properties and composition.
- Substances can be classified as elements or compounds.
- Compounds can be simplified (separated) into simpler substances through chemical reactions. Elements cannot be simplified.
- Mixtures can be simplified by physical changes.
the law of conservation of mass
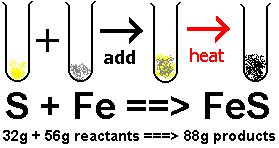 Law of Conservation of Mass
Law of Conservation of Mass
"Mass is neither created nor destroyed in any chemical reaction."
Matter may change is forms in a variety of ways, there is never a loss in total mass.
WATCH Law of Conservation of Mass
WATCH Antoine Lavoisier and the Law of Conservation
WATCH NOVA "Hunting the Elements"
Matter may change is forms in a variety of ways, there is never a loss in total mass.
WATCH Law of Conservation of Mass
WATCH Antoine Lavoisier and the Law of Conservation
WATCH NOVA "Hunting the Elements"