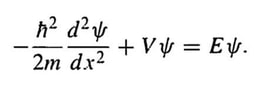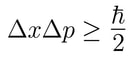KEY IDEA: Electrons in atoms adopt stable orbits at specific energies.
- The behavior of electrons can best be described as wavelike in character and occupy discrete energies within the atom.
- With a few simple rules, the electron configuration of each element can be described as four quantum numbers.
Development of atomic models
|
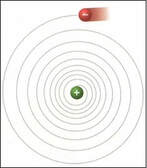
Rutherford’s atomic model - light, negatively charged electrons orbiting a dense, positively charged nucleus - seems physically impossible. In his model, the electrons occupy an undefined space held in place by electrostatic attraction. Each electron is indistinguishable from the others; they only balance the positive charge at the center of the atom.
Classical mechanics predicts the orbiting electrons would spiral into the nucleus. Rutherford's model also couldn't explain why metals change color when they're heated up. |
Questions remain... What holds the atom together? How do atoms interact with light?
the bohr model
|
In 1913 Neils Bohr proposed an improvement on the Rutherford model; his model overcame classical mechanics and paved the way for quantum mechanics. Although Bohr's model has been replaced by a more accurate, but complex, quantum mechanical-model, its underlying principles are valid.
|
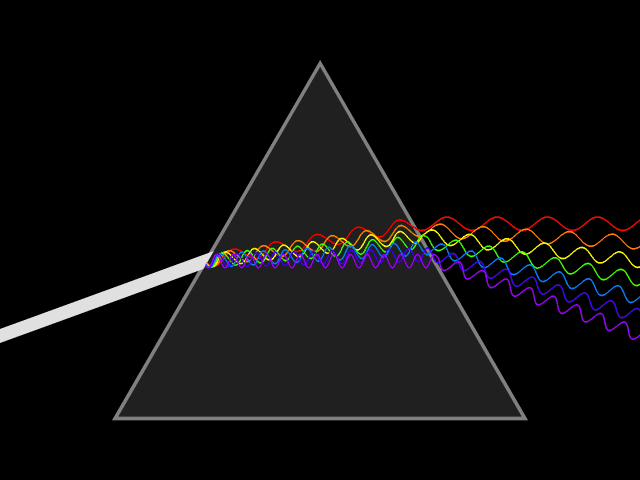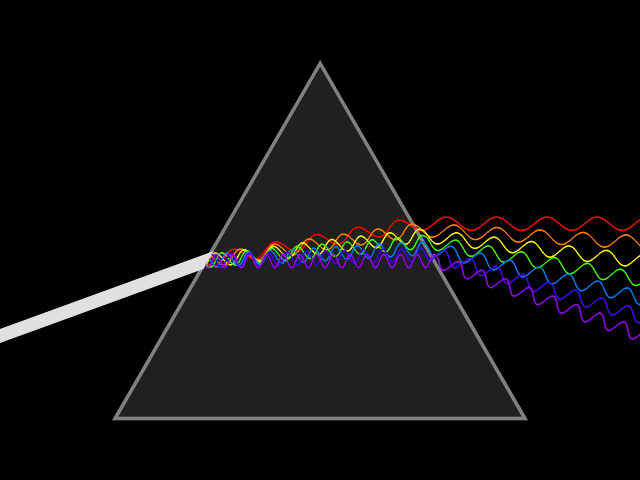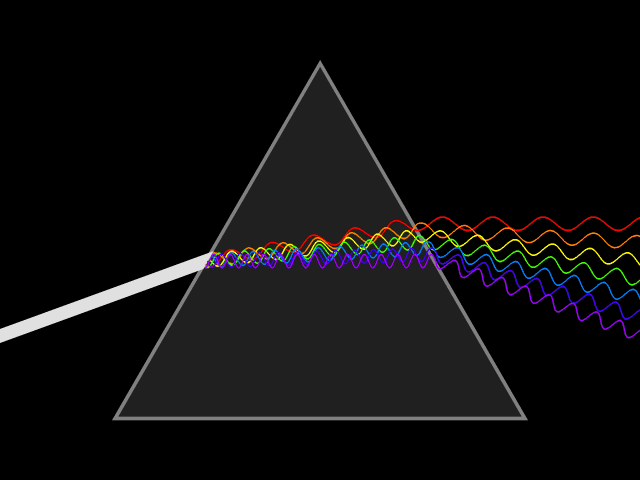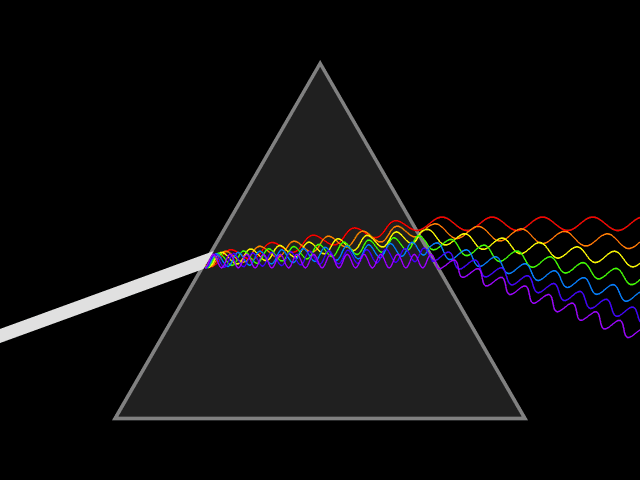
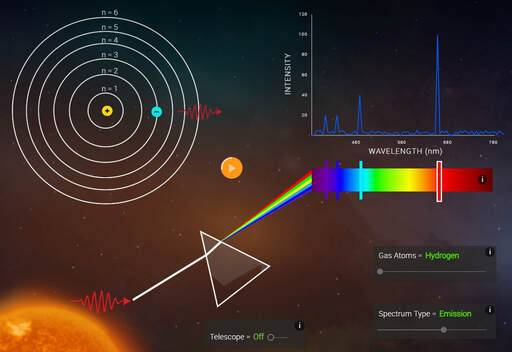
Bohr developed his model after studying the way glowing hydrogen gives off light.
When the light produced by hydrogen gas is passed through a prism, only a few specific wavelengths are seen.
Bohr said these specific wavelengths suggested that electrons adopt specific energies, and that the spectral lines are the result of transitions between energy levels. The color of the spectral line corresponds to photons of a frequency related to the energy difference between levels.
LINK: Spectrum of Hydrogen Atom (Java Lab)
Bohr said these specific wavelengths suggested that electrons adopt specific energies, and that the spectral lines are the result of transitions between energy levels. The color of the spectral line corresponds to photons of a frequency related to the energy difference between levels.
LINK: Spectrum of Hydrogen Atom (Java Lab)
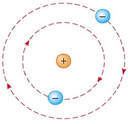
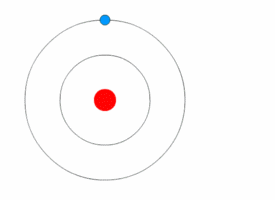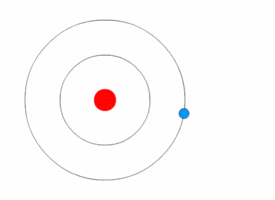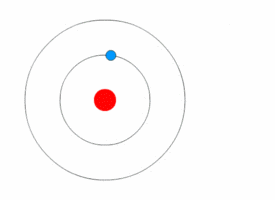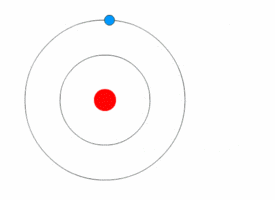
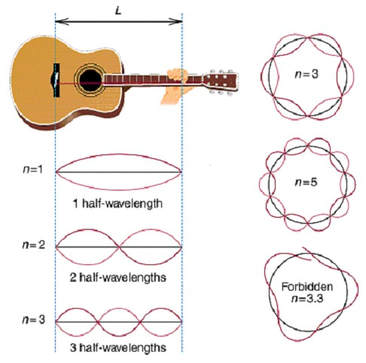
In Bohr's atomic model, the electrons can reside at discrete, well-defined distances from the nucleus. These "energy states" are similar to the rungs of a ladder:
|
You can stably stand on any one step, but not anywhere in between two steps.
Only certain specific distances from the floor are possible, or "allowed." And since each step has a different gravitational potential energy; only certain specific energies on the ladder are allowed. In an atom, different electron orbits have specific electromagnetic potential. The electron can reside in only one state without accelerating. Light is emitted only when an electron jumps from a high energy state to a lower energy state. The lowest energy orbit is called the "ground state"; all higher energy orbit are called "excited states." |
VIDEO: Photoemission Spectra
Main ideas of Bohr's atomic model:
- Energy and light must come in discrete packets, called "quanta."
- The smallest increment in energy is represented by an electron's jump from one orbit to the next - called a quantum jump.
- A quantum jump from a high energy level to a lower energy level releases one quantum of electromagnetic radiation, called a photon. When an electron absorbs a photon, an electron jumps to a higher energy orbit.
The Bohr model was successful in modeling the behavior of simple atoms and their interactions with light. But it did not explain more complex atoms. It's too simple.
Today's atomic model is based upon the principles of wave mechanics. The basic ideas are complicated and involve complex mathematics. Some of the conclusions of this theory, however, help explain the arrangement of electrons in atoms.
Today's atomic model is based upon the principles of wave mechanics. The basic ideas are complicated and involve complex mathematics. Some of the conclusions of this theory, however, help explain the arrangement of electrons in atoms.
Electromagnetic Radiation and light
Electromagnetic radiation is composed of waves that travel at the speed of light (c).
c = 299,792.458 km/sec
(or 3.00 × 10^8 meters per second)
(or 3.00 × 10^8 meters per second)
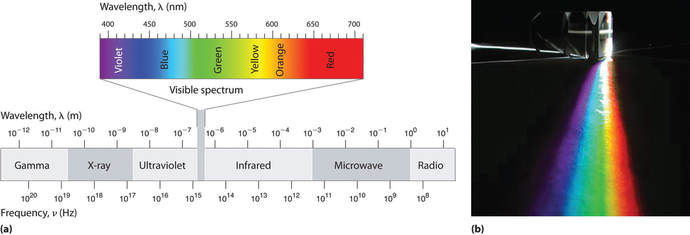
Electromagnetic radiation makes up the electromagnetic spectrum which covers a wide range of wavelengths and frequencies, (a), from low frequency (long wavelength) radio waves through visible light and into high frequency (short wavelength) cosmic ray region. The human eye is sensitive to a small portion of the electromagnetic spectrum, visible light (b).
MEDIA GALLERY: Explore the basic characteristics of waves--amplitude, frequency, and wavelength—and see how they correspond with observations of light and sound.
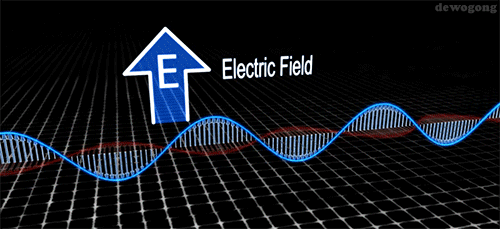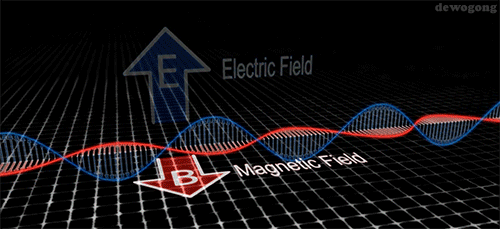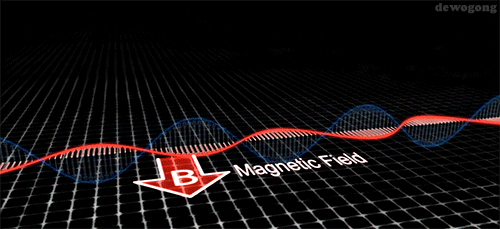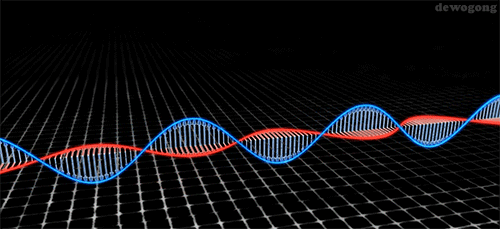
Electromagnetic waves are composed of an electric field and a magnetic field:
Electromagnetic waves are composed of an electric field and a magnetic field:
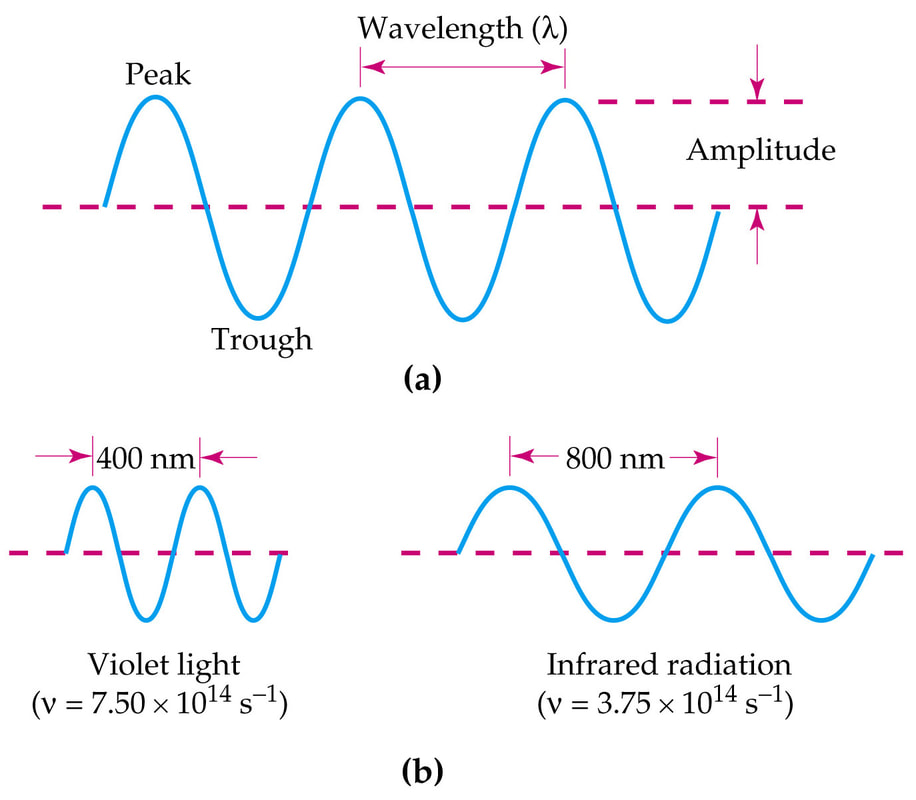
Waves are described by their amplitude, wavelength, and frequency:
|
The brightness of light depends on the amplitude, or height, of the light wave.
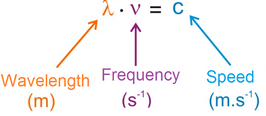
(a) The wavelength (λ, "lambda") is the distance of one complete wave. Wavelength is commonly measured in nanometers
(b) The frequency (v, "nu") is measured as the number of complete wavelengths per second. The relationship between wavelength and frequency is given by: |
Note that wavelength and frequency are inversely proportional.
VIRTUAL LAB: Electromagnetic Waves
APPLET: Transverse Traveling Wave
VIRTUAL LAB: Electromagnetic Waves
APPLET: Transverse Traveling Wave
quantum theory
|
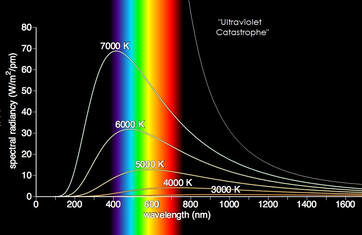
The idea that electromagnetic waves could be quantized was first proposed in 1900 by the German physicist Max Planck to explain the spectrum of radiation emitted by a blackbody. An ideal blackbody, such as iron, absorbs all incoming light and does not reflect any. Most of this energy is re-radiated into the surrounds as heat. At room temperature, such an object would appear black, hence blackbody.
When heated, a black body begins to emit red and then orange light, and eventually white-hot at higher temperatures. Classical mechanics overestimates the amount of energy that would be radiated at very short wavelength; a problem that came to be known as the "ultraviolet catastrophe." |
To solve this problem, Planck suggested that light-energy does not follow classical theory, but comes in discontinuous steps, or quanta. In other words, energy is contained in multiples of a fundamental unit. The energy (E) carried by a photon of a light wave is given by the wave's frequency times a constant, now known as Planck's constant:
WATCH The Ultraviolet Catastrophe (Physics Girl)
WATCH The Ultraviolet Catastrophe (Physics Girl)
WATCH The Origin of Quantum Theory (Britannica)
the Photoelectric effect
|
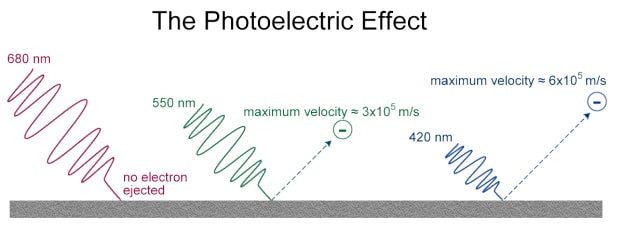
In 1905, Albert Einstein used Planck's quantum theory to explain the photoelectric effect - shining light on a metal surface causes the metal to emit electrons.
For each metal, there is a minimum frequency of light below which no electrons are emitted. Einstein explained this effect as a result of light energy being carried in discrete quantized packets, called photons. These particles of light are concentrations of energy incorporated into the wave. |
wave-particle duality
|
Einstein's introduction of particles of light suggested that particles, like an electron, have properties of a wave. In 1924, French physicist Louis de Broglie extended wave nature to all matter, thereby allowing use of wave mechanics to describe the behavior of electrons in atomic systems.
WATCH What is the Wave/Particle Duality? (part 1) WATCH The Wave/Particle Duality (part 2) |
The wave-mechanical picture of atomic structure is one that considers the electrons to have wavelike character.
TED-Ed "Is light a particle or a wave?"
TED-Ed. "Particles and waves: The central mystery of quantum mechanics"
TED-Ed. "Particles and waves: The central mystery of quantum mechanics"
schrödinger's wave equation
Schrödinger claimed his wave equation describes a real physical picture of the atom with electrons as waves surrounding the atomic nucleus.
Heisenberg Uncertainty Principle
|
German physicist Werner Heisenberg joined Bohr in 1926. Heisenberg realized that the act of measuring an electron's properties would alter the electron's behavior. The act of observing disturbs the observed.
Furthermore, the position and the velocity of an object cannot both be measured exactly, at the same time, even in theory. The more precisely you measure one property, the less precisely you can determine the other. This idea is tied up in an equation using Planck's constant, and called it the uncertainty principle. |
Heisenberg had uncovered a deep and shocking truth about the atomic world. Atoms are willfully obscure. We can never fully know an atom's position and speed simultaneously.

TED-Ed. "Schrödinger's cat: A thought experiment in quantum mechanics"
TED-Ed. What can Schrödinger's cat teach us about quantum mechanics?
TED-Ed. What is the Heisenberg Uncertainty Principle?
The Double-Slit Experiment (YouTube)
TED-Ed. What can Schrödinger's cat teach us about quantum mechanics?
TED-Ed. What is the Heisenberg Uncertainty Principle?
The Double-Slit Experiment (YouTube)
|
WATCH Quantum Theory Made Easy
The course breaks down the concepts of quantum physics into a digestible manner. More in-depth than most presentations for laypersons, but without the mathematical rigor required for a specialist in the field. (31 min.) |
quantum mechanical model
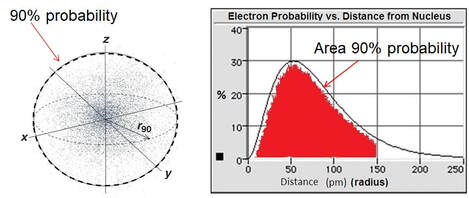
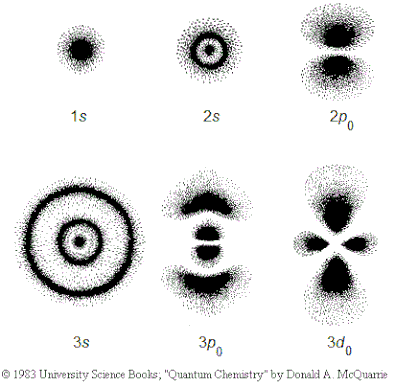
Modern atomic theory describes the electronic structure of the atom as the probability of finding electrons within certain regions of space.
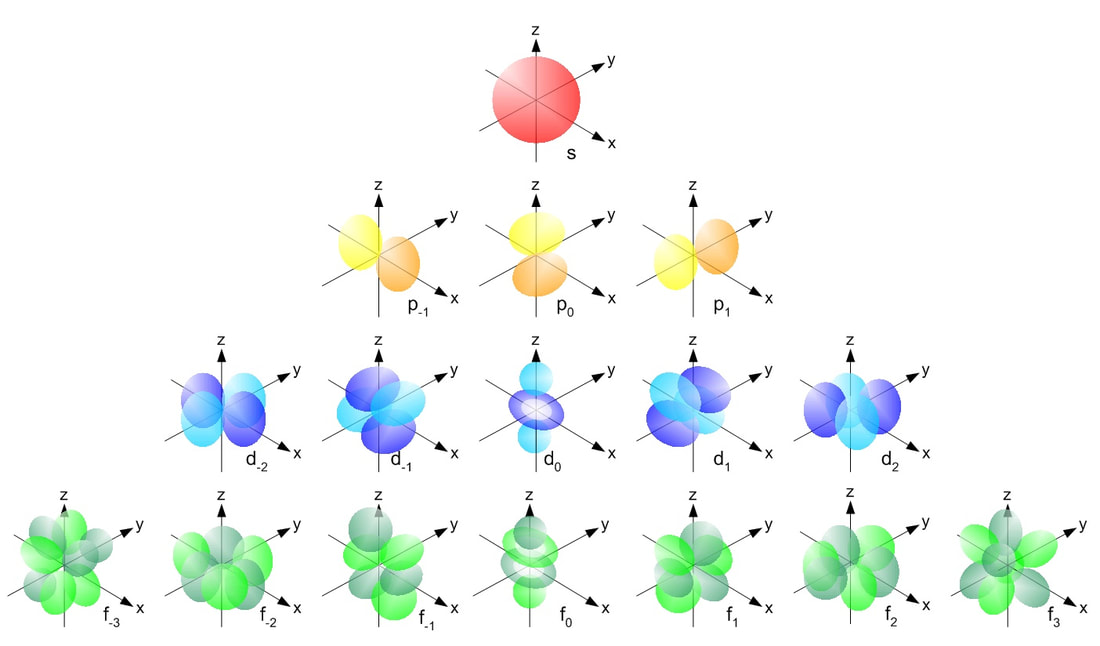
ATOMIC ORBITAL
A region of space where there is a high probability of finding an electron.
A region of space where there is a high probability of finding an electron.
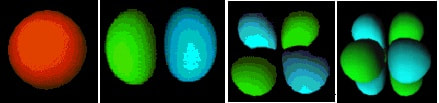
There are 4 basic orbital shapes: s = sphere; p = peanut; d = diamond; f = flower
The orbitals are 3-dimensional, and can be orientated in different ways:
These orbital shapes represent the regions of space in which there is a high probability of finding an electron.
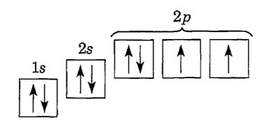
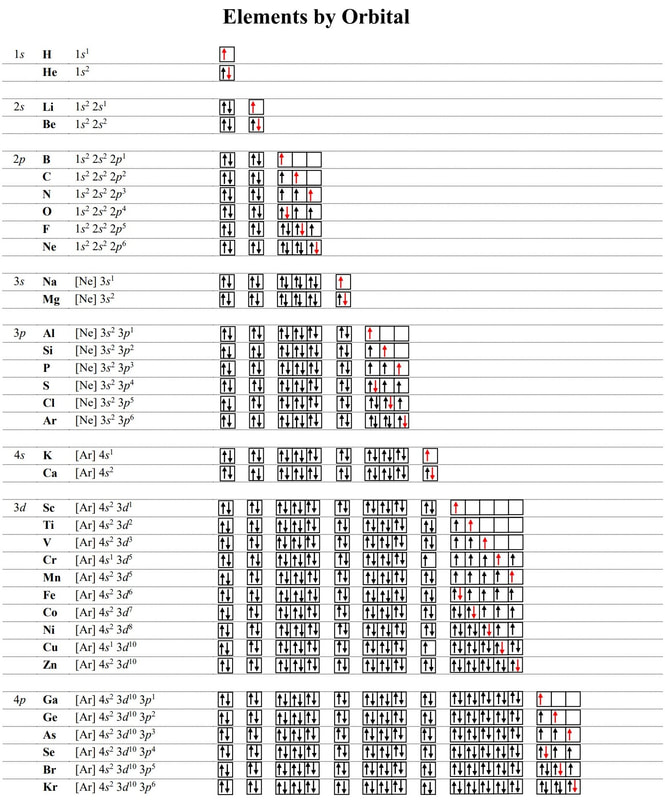
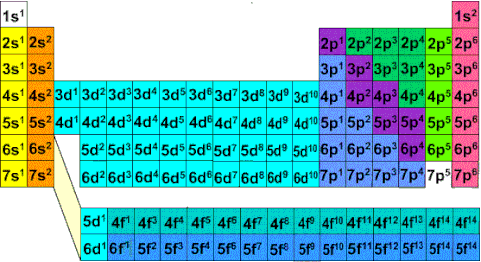
Electron-configurations & orbital diagrams
"Exotic" orbitals beyond f-sublevels exist theoretically, and may be populated in some excited states.
ELECTRON-CONFIGURATION
Electron configuration is a way of describing the location of electrons in an atom.
Electron configuration is a way of describing the location of electrons in an atom.
Three General Rules:
1. Pauli Exclusion Principle
1. Pauli Exclusion Principle
- Each orbital can hold TWO electrons with opposite "spin."
- Orbitals are occupied by single electrons before pairing.
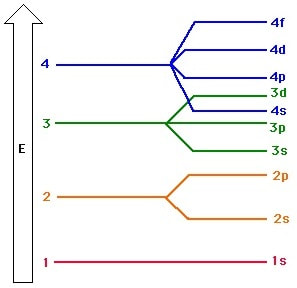
- Electrons fill the lowest energy orbitals first.
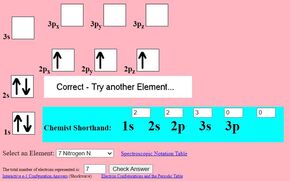
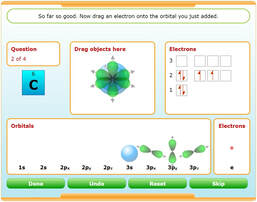
Electron-Configuration Interactives
Use these activities to build up orbitals with electrons and construct elements.
Use these activities to build up orbitals with electrons and construct elements.
There are exceptions to the aufbau principle with the d-block elements chromium, Cr, and copper, Cu. Stabilization is achieved with a complete and symmetric set of five 3d orbitals containing 5 or 10 electrons. Thus,
- Chromium has the configuration: [Ar] 3d5 4s1 - not: [Ar] 3d4 4s2
- Copper has the configuration: [Ar] 3d10 4s1 - not: [Ar] 3d9 4s2
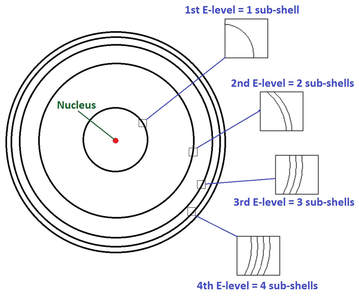
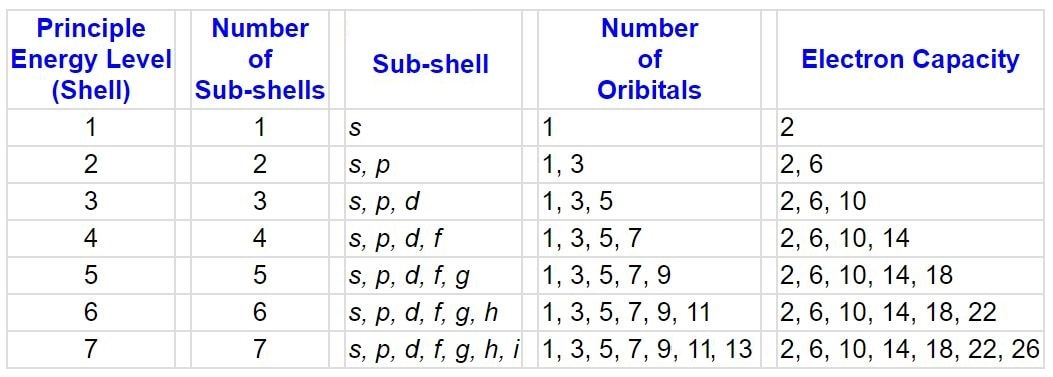
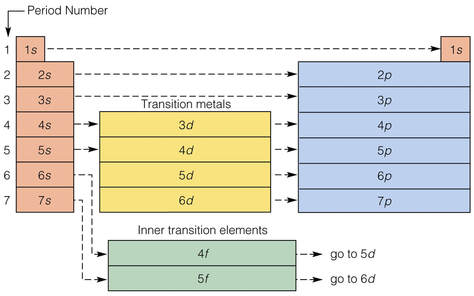
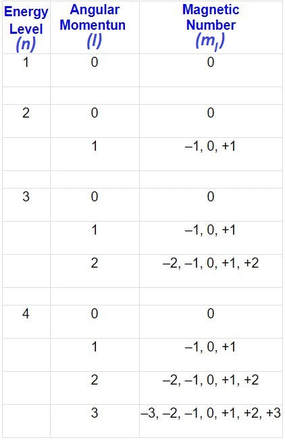
four (4) quantum numbers
The behavior of electrons can be summarized by 4 quantum numbers, which designate the energy and location of electrons in electron orbitals:
1. The principle quantum number (n) specifies an electron's energy level, and determines the size of the orbital
2. The angular momentum quantum number (l = lowercase letter "L") indicates the orbital type. (s, p, d, or f)
3. The magnetic quantum number denotes the spatial orientation of an orbital.
4. The spin quantum number determines the electron spin.
1. The principle quantum number (n) specifies an electron's energy level, and determines the size of the orbital
- n can only have values of 1, 2, 3, ...
2. The angular momentum quantum number (l = lowercase letter "L") indicates the orbital type. (s, p, d, or f)
- l can only have values of 0, 1, 2, 3, ... (n – 1)
- s = 0; p = 1; d = 2; f = 3
3. The magnetic quantum number denotes the spatial orientation of an orbital.
- ml can have values from –l to +l
4. The spin quantum number determines the electron spin.
- ms can have values of +1/2 or –1/2
- electrons spin on an axis, thereby generating a magnetic field
Consider the e-configs and orbital diagrams for the elements in the second period of the periodic table:
|
The 4 quantum numbers for the electron
indicated red in the orbital diagram: Li: n = 2, I = 0, ml = 0, ms = +1/2 Be: n = 2, I = 0, ml = 0, ms = –1/2 B: n = 2, I = 1, ml = –1, ms = +1/2 C: n = 2, I = 1, ml = 0, ms = +1/2 N: n = 2, I = 1, ml = +1, ms = +1/2 O: n = 2, I = 1, ml = –1, ms = –1/2 F: n = 2, I = 1, ml = 0, ms = –1/2 Ne: n = 2, I = 1, ml = +1, ms = –1/2 |
Quantum numbers can be written for any electron within an atom. No two electrons can have the same four quantum numbers.
|
Explore further...
WATCH Feynman's Story of Particle Physics WATCH What is the Bohr model of the atom? WATCH BBC's Atom: Clash of Titans (pts. 2 & 3) WATCH NOVA: Fabric of the Cosmos, "Quantum Leap" WATCH Quantum Entanglement: Atomic Physics and Reality WATCH Michio Kaku "The Universe in a Nutshell" (full presentation) WATCH Quantum Mechanics TED-Ed Watchlist VIDEO COURSE Quantum Mechanics (Stanford Univ.) eBOOK USCD Physics 130 |