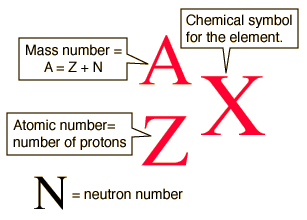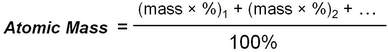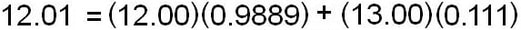KEY IDEA: Atoms are submicroscopic particles that make up all things.
|
Ideally, grade school students have been gradually introduced to atomic theory and covered basic concepts in the structure of atoms. By high school, they should be familiar with major aspects of the theory.
WATCH Voyage into the world of atoms |
define the atom
|
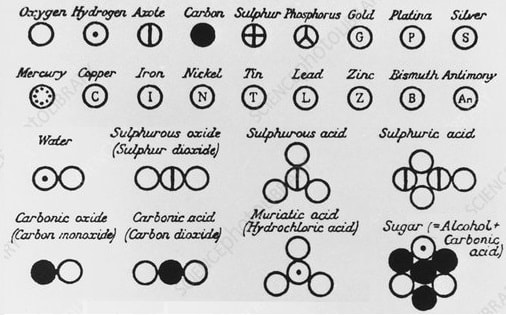
Since ancient times, philosophers have asked, "What are we made of?" Is there a more simple basic "building block" common to ALL matter? Many believed the great variety in matter is made of the same thing.
Democritus (c.460–370 BC) - Greek philosopher
|
|
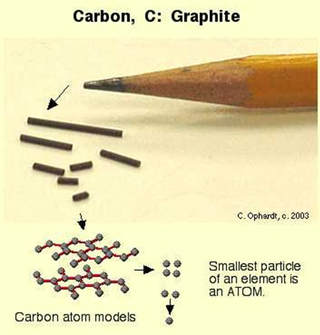
Take the example of a piece of graphite, which is carbon.
The graphite can be cut in half to make two smaller pieces of graphite. Those can be cut into smaller pieces as well, and they are still graphite. This process can continue creating ever smaller and smaller samples of that element: carbon. But if we could continue to do this perpetually ... eventually we will arrive at a point where we have just one atom of carbon left in our sample. |
We now know atoms are divisible. But the fragments created by splitting an atom of carbon are no longer carbon; they’re something else.
Atom is the term that refers to the smallest particle of an element that retains the identify of that element.
Atom is the term that refers to the smallest particle of an element that retains the identify of that element.
Till about 250 years ago, the existence of atoms was a philosophical debate. By the mid-1700s, scientists finally figured out ways to experiment directly with atoms.
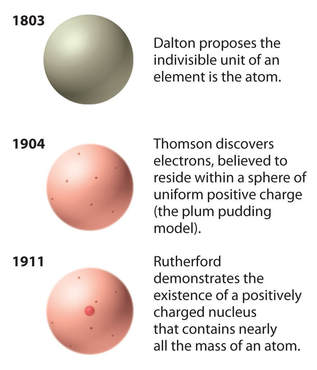
dalton's atomic theory
|
1805 - John Dalton - English meteorologist, presented a summary of Atomic Theory:
Dalton's model of the atom is indivisible with no internal structure. (Like a glass marble.) |
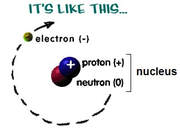
atomic structure
Dalton was not correct in some parts of his theory: atoms are divisible.
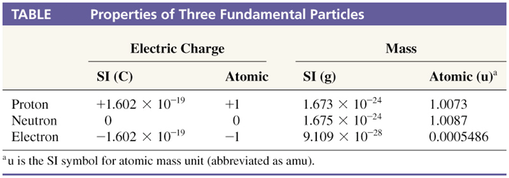
Summary of atomic particles:
Summary of atomic particles:
|
location
|
relative charge
|
relative mass
|
|
proton
|
in the nucleus
|
+1
|
1
|
|
electron
|
orbiting the nucleus
|
–1
|
0 *
|
|
neutron
|
in the nucleus
|
no charge
|
1
|
* mass of electron = 1/1840 mass of proton
Protons and neutrons are collectively called "nucleons" - subatomic particles found in the nucleus.
Historical DevelopmentObservations that led to the nuclear model were discovered over several years in the 1800's.
Critical experiments: |
|
Cathode Ray Tube, "CRT"
1897 - J.J. Thomson discovered that a magnet deflects the invisible current and concluded that the beam has an electric charge and mass. |
1909 - Robert Millikan measured the electron's mass and charge in his famous "oil-drop" experiment.
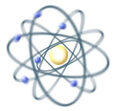
1909 - Ernest Rutherford concluded that the atom is almost entirely empty space, with a tiny nucleus that carries a positive charge and almost all of the atom’s mass. Around this nucleus orbit the negatively charged electrons.
This model led to the discovery of the radioactive elements.
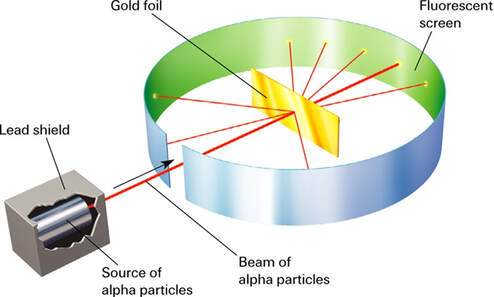
Rutherford's "gold-foil" experiment:
A beam of "alpha particles" (produced in the radioactive decay of uranium) aimed at a piece of gold foil.
Most of the alpha particles passed through the gold, but some were scattered at high angles and, on occasion, deflected backwards.
A beam of "alpha particles" (produced in the radioactive decay of uranium) aimed at a piece of gold foil.
Most of the alpha particles passed through the gold, but some were scattered at high angles and, on occasion, deflected backwards.
Rutherford is famously quoted as saying, "It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you."
In the following simulation, explore the differences in the experimental predictions of each model of the atom.
In the following simulation, explore the differences in the experimental predictions of each model of the atom.
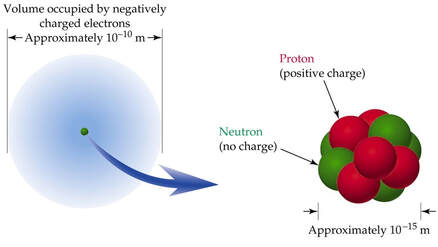
Rutherford concluded that atoms were mostly empty space, with a highly concentrated nucleus of positive charge which contain most of its mass in just a small fraction of the atom’s total volume.
Atoms have diameters of around 100 picometers, or about one‑tenth of a nanometer. Rutherford’s work eventually led to the discovery that the radius of a typical nucleus is only about 1/100,000th that of its electron cloud.
That is, 1 femtometer, or 0.000001 nanometers.
That is, 1 femtometer, or 0.000001 nanometers.
"Man is equally incapable of seeing the nothingness from which he emerges and the infinity in which he is engulfed.” -Blaise Pascal
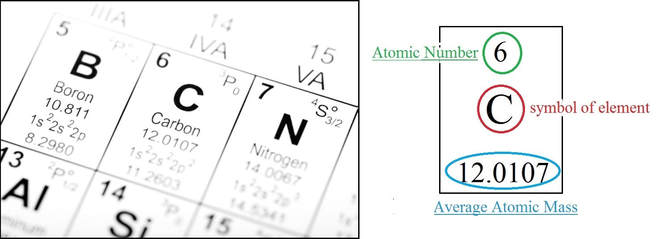
atomic symbols
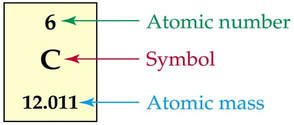
ATOMIC NUMBER
- "nuclear charge:" the number of protons in the nucleus of an atom
- unique to each element; sequential on periodic table
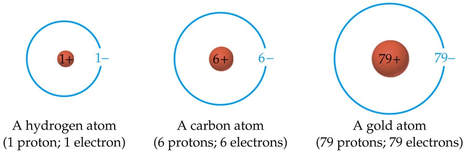
- atoms are neutral; the number of protons equals the number of electrons
Rutherford named the hydrogen nucleus, "proton" (Greek for "first").
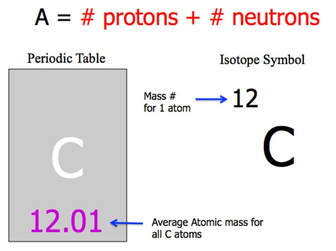
MASS NUMBER
- the total number of protons plus neutrons (always a whole-number)
- mass of an electron is negligible, and thus ignored.
- Average-Atomic mass: the average mass of all atoms of that element (typically a decimal-number)
NUCLEAR NOTATION
Meanwhile...
1913 - T. W. Richards found two atomic masses for lead.
1919 - F. W. Aston invented mass spectrograph and separated neon atoms into two different masses.
1921 - Frederick Soddy earned the Nobel Prize for his investigations into the origin and nature of isotopes.
1913 - T. W. Richards found two atomic masses for lead.
1919 - F. W. Aston invented mass spectrograph and separated neon atoms into two different masses.
1921 - Frederick Soddy earned the Nobel Prize for his investigations into the origin and nature of isotopes.
The Mass Spectrometer: How it works, and example calculation.
ISOTOPES
- atoms that have the same atomic number, but different mass number
- atoms of the same element with different numbers of neutrons
Many isotopes of the elements have been discovered.
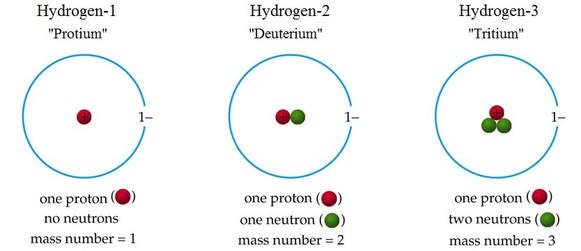
There are 3 isotopes of hydrogen:
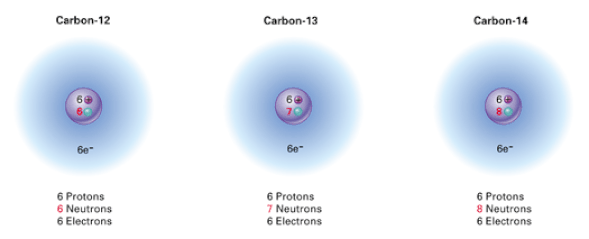
There are 3 isotopes of carbon:
The most common form of carbon is the carbon-12 isotope:
The atomic mass for an element is a weighted average of all isotopes of that element:
The atomic mass is calculated using the percent abundances of isotopes
For example, to calculate the atomic mass of carbon, use the mass and percent abundances given above: (ignoring C-14)
|
"Arguably, the single greatest scientific discovery is that everything is made of atoms. The infinite variety and forms of matter comes down to tiny atoms and the mysterious laws they obey. As scientists unraveled the structure of atoms, and begin to look into the very heart of matter, they learned nature's most shocking secrets. They had to abandon everything they believed and create a whole new science, which today underpins the entire universe and even life itself."
|
Explore further...
LINK Elements & Atoms: Cases Study in the Development of Chemistry
LINK Symmetry Magazine
WATCH Standard Model of Particle Physics
WATCH How Small Is It?
LINK Elements & Atoms: Cases Study in the Development of Chemistry
LINK Symmetry Magazine
WATCH Standard Model of Particle Physics
WATCH How Small Is It?