KEY IDEA: Chemistry is a quantitative science.
- Chemists must be able to make accurate and precise measurements.
- Measurements should be reported by a common set of units and reflect the quality of the data.
- Problem solving is made easy by use of dimensional analysis.
Measurement
Scientific notation
LINK The Scale of the Universe LINK Secret Worlds: The Universe Within LINK A Tediously Accurate Map of the Solar System |
Accuracy vs. Precision
The "quality" of a measurement is described in terms of its accuracy and its precision:
Accuracy is how close a single measurement is to the accepted (or "true") value.
Precision is how close a series of measurements are to each other.
Accuracy is how close a single measurement is to the accepted (or "true") value.
- compare a single measurement to the "correct" value
Precision is how close a series of measurements are to each other.
- compare multiple measurements to each other
All measurements, no matter how accurate, contain some error.
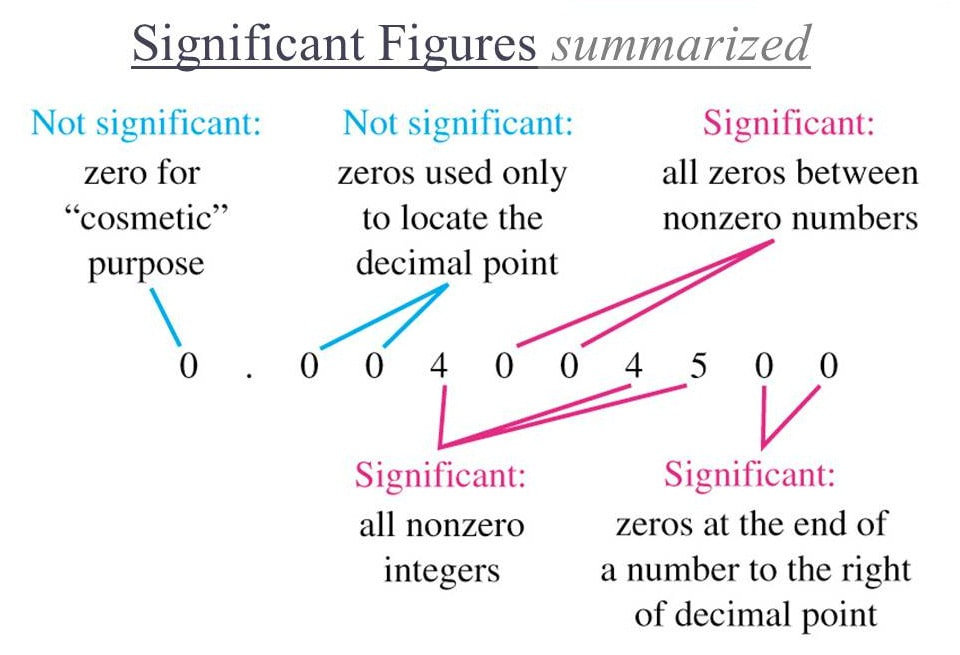
significant figures
|
ALL non-zero numbers are significant.
Zeros may or may not be significant:
|
QUIZ Sig-Figs
"SIG-FIGS" IN CALCULATIONS:
A measurement is only as precise as the numbers used to calculate it.
A measurement is only as precise as the numbers used to calculate it.
- When multiplying or dividing, the number with the fewest sig-figs determines the number of sig-figs.
- When adding or subtracting, the number with the lowest decimal value determines the number of sig-figs.
international system of units
A coherent system of units grew out of a need for standard and reproducible measurements for science and commerce. Between 1791-1795, the French Academy of Sciences developed the metric system, based on decimal multiples.
In 1960, an international organization was drawn up to develop and coordinate a world-wide uniform system of measurements, the International System of Units, or "SI." The SI system declares only a minimum number of fundamental units, rigorously defined in terms of reproducible phenomena or objects. All other units are derived from combinations of these base units.
In 1960, an international organization was drawn up to develop and coordinate a world-wide uniform system of measurements, the International System of Units, or "SI." The SI system declares only a minimum number of fundamental units, rigorously defined in terms of reproducible phenomena or objects. All other units are derived from combinations of these base units.
|
Base Unit
Derived Unit
|
|
Prefixes are used to make multiples of the base unit:
NOTE: There is one "correct" unit, but many equivalent prefixes of that unit. For example, you'd use meters to measure your height, but you could use centimeters or millimeters. |
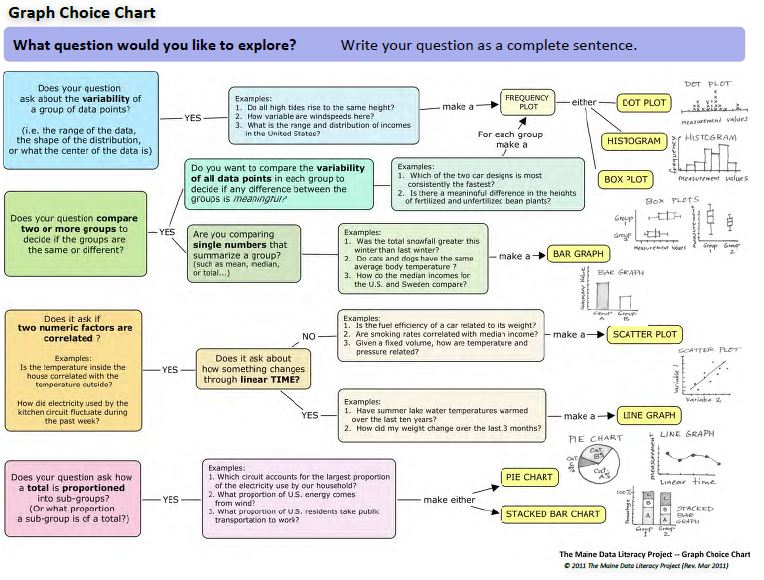
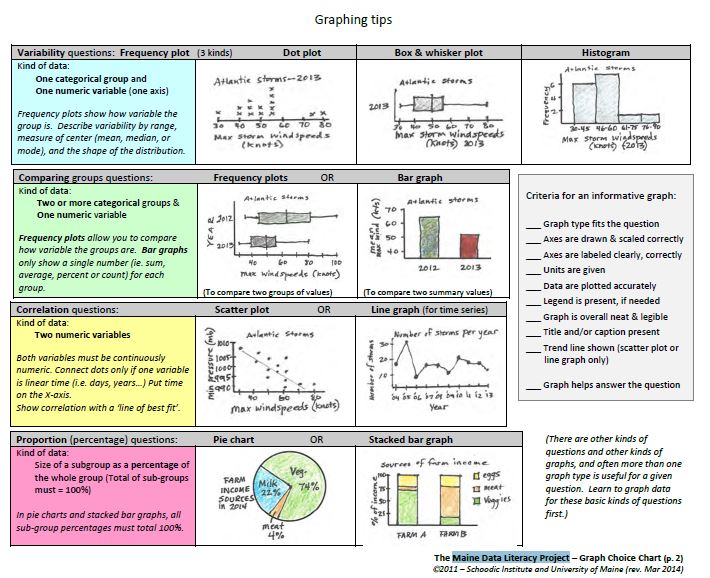
GRAPHING
Constructing and interpreting graphs is an important skill, not only in the area of science, but also geography and mathematics. In the real world, graphs are used on a daily basis to communicate important information.
dimensional analysis
- A way of converting between different units of measurements.
A conversion factor relates two units as a numerical equivalence, and can be used to translate between different systems of measurement.
In dimensional analysis, a measurement is multiplied by one or more conversion factors to express that measurement in different units.
For example: A standard marathon is 26.22 miles long. How many kilometers is this? (Given, 1 km = 0.6214 mi)
For example: A standard marathon is 26.22 miles long. How many kilometers is this? (Given, 1 km = 0.6214 mi)
Dividing the original measurement (in miles) by 0.6214 cancels the unit miles, and leaves the desired unit, kilometers.
To convert FROM inches (in) TO centimeters (cm), given 1 inch equals 2.54 centimeters:
Multiplying a measurement in inches by 2.54 cancels the unit inches, and leaves the desired unit, centimeters.
To convert FROM centimeters TO inches, the reciprocal conversion factor can be used:
To convert FROM centimeters TO inches, the reciprocal conversion factor can be used:
When a single conversion doesn't exist, the conversion can be made by multiple conversions, applied in sequence.
When converting Liter to cups:
Dimensional analysis can be used converting metric prefixes. For example, 1 mm equals 1/1000th of a meter:
Squared units can be calculated by squaring the equivalence. To convert between square inches and square centimetres:
The same equivalence is cubed for converting from cubic centimeters to cubic inches:
Density is an important physical property relating the mass of an object to its volume. Density can be used as a conversion between units of mass and units of volume for a specific substance
For example, the density of a substance is given as 1.32 g/cm^3. To convert from grams to milliliters:
A cubic centimeter is the same volume as a milliliter. (1 cm^3 = 1 mL)
To convert from kilograms to liters, using the density given 0.789 g/cm^3.
To convert from kilograms to liters, using the density given 0.789 g/cm^3.
|
Unit conversion tables (good source for conversions)
|
QUESTION: Calculate the mass in grams of a 1.25 in^3 sample of osmium. The density of osmium is 22.48 g/cm^3. (ANSWER)