KEY IDEA: Stoichiometry is the study of the mass relationships between substances in a chemical reaction.
- A balanced chemical equation is the key to solving stoichiometry problems.
Stoichiometry
"stoy-key-om-a-tree"
Stoichiometry is the quantitative study of chemical reactions. (Stoicheon: element; metron: measure)
The coefficients in a balanced chemical equation give the relative amounts of substances involved in a chemical reaction.
Consider the formation of ammonia (NH3) from the combination of nitrogen and hydrogen:
The coefficients in a balanced chemical equation give the relative amounts of substances involved in a chemical reaction.
Consider the formation of ammonia (NH3) from the combination of nitrogen and hydrogen:
Only atoms and mass are conserved in a chemical reaction. (Dalton's Atomic Theory, Lavoisier's Law of Conservation.)
|
The above reaction is called the "Haber Process," in honor of German chemist Fritz Haber for his method. (TED-Ed.)
|
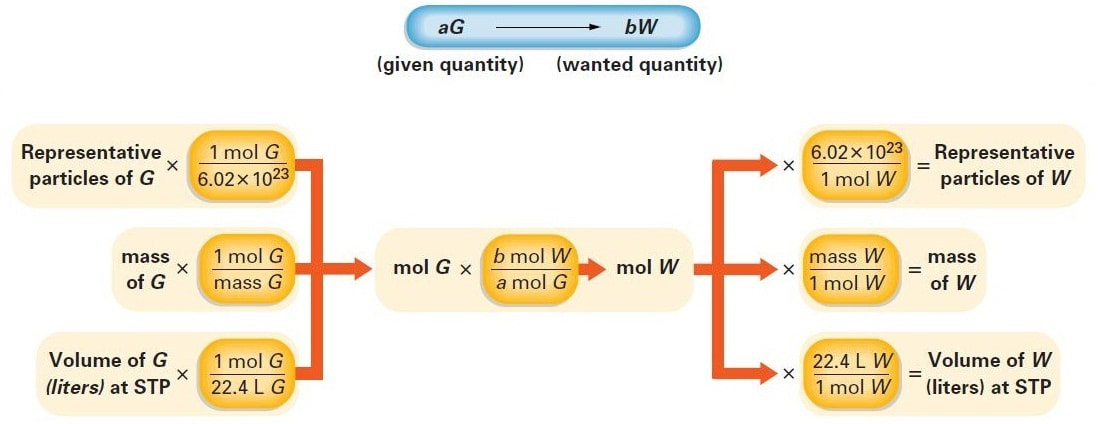
The following figure shows the calculation for a general "mass-to-mass" stoichiometric conversion:
The factors can be adapted to any mass-mass conversion:
Recall dimensional analysis. ALL stoichiometry problems can be solved by the "factor-label" method.
- The mass-moles factor can be found from a periodic table.
- The balanced equation provides the mole-mole factor.
Recall dimensional analysis. ALL stoichiometry problems can be solved by the "factor-label" method.
The following figure shows the steps for solving a variety of stoichiometry calculations:
Stoichiometry Made Simple This interactive provides step-by-step explanations and examples of many different types of stoichiometry problems. (Genially.com)
Limiting & Excess Reactants
|
After a chemical reaction, typically one reactant gets used up, while another reactant is left over.
The reactant that gets used up first is called the "limiting reactant" (or "limiting reagent.") The limiting reactant determines the maximum amount of product formed. Once all the limiting reactant is used up in a chemical reaction, the reaction stops and no more product can be formed. |
The following "recipe" calls for 3 moles of H2 for every mole of N2:
In this particular experiment, H2 is the "limiting reagent" and N2 is the "excess reagent".
Sample calculation:
Determine the amount of ammonia (NH3) produced when 45.8 grams of nitric oxide (NO) are mixed with 12.4 grams of hydrogen gas, according to the equation:
Determine the amount of ammonia (NH3) produced when 45.8 grams of nitric oxide (NO) are mixed with 12.4 grams of hydrogen gas, according to the equation:
The theoretical yield of ammonia is 26.0 grams, as determined by the limiting reactant.
|
Need help?
|